|
 |
| J Korean Ophthalmol Soc > Volume 62(7); 2021 > Article |
|
ĻĄŁļ¼Ėņ┤łļĪØ
ļ¬®ņĀü
ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Ø ĒÖśņ×ÉņŚÉņä£ ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņ×ģņłĀĻ│╝ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀ Ļ░äņØś ņ╣śļŻī ĒÜ©Ļ│╝ ņ░©ņØ┤ļź╝ ņĢīņĢäļ│┤Ļ│Āņ×É ĒĢ£ļŗż.
ļīĆņāüĻ│╝ ļ░®ļ▓Ģ
ņ▓śņØī ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Øņ£╝ļĪ£ ņ¦äļŗ©ļ░øĻ│Ā ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ĒĢŁĒśłĻ┤Ćļé┤Ēö╝ņä▒ņןņØĖņ×É ņŻ╝ņé¼ļź╝ ļ░øņØĆ ĒÖśņ×É 49ļ¬ģ 51ņĢłņØä ļīĆņāüņ£╝ļĪ£ ĒĢśņŚ¼ ņØśļ¼┤ĻĖ░ļĪØņØä ĒøäĒ¢źņĀüņ£╝ļĪ£ ļČäņäØĒĢśņśĆļŗż. ĒĢŁĒśłĻ┤Ćļé┤Ēö╝ņä▒ņןņØĖņ×É ņŻ╝ņé¼ ņóģļźśņŚÉ ļö░ļØ╝ ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖņØä ņŻ╝ņ×ģĒĢ£ ĻĄ░Ļ│╝ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖļź╝ ņŻ╝ņ×ģĒĢ£ ĻĄ░ņ£╝ļĪ£ ĻĄ¼ļČäĒĢśņśĆĻ│Ā, ņŗ£ņłĀņŚÉ ļīĆĒĢ£ ļ░śņØæņØ┤ ņŚåĻ▒░ļéś ņĢģĒÖö ņŗ£ ļÅÖņØ╝ĒĢ£ ņĢĮņĀ£ļĪ£ ļ░śļ│Ą ņ╣śļŻīĒĢśņśĆļŗż. ņ╣śļŻī ņØ┤Ēøä 3Ļ░£ņøö ņØ┤ņāüņØś ņČöņĀü Ļ┤Ćņ░░ņØä ņŗ£Ē¢ēĒĢśņśĆĻ│Ā, ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀźĻ│╝ ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗ś ļ│ĆĒÖö, ņ╣śļŻīĻ╣īņ¦Ć ņŗ£Ē¢ēļÉ£ ņŻ╝ņé¼ Ēܤņłś ļ░Å ĻĖ░Ļ░äņØä ļČäņäØĒĢśņśĆļŗż.
Ļ▓░Ļ│╝
ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņ×ģņłĀĻ│╝ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀ ļ¬©ļæÉ ņ╣śļŻī ņĀäĒøä ņØśļ»Ė ņ׳ļŖö ņŗ£ļĀźļ│ĆĒÖö(p<0.0001, p=0.001)ņÖĆ ņżæņŗ¼ļ¦źļØĮļ¦ē ļæÉĻ╗ś ļ│ĆĒÖöļź╝ ļ│┤ņśĆļŗż(p<0.0001, p=0.011). ĒĢśņ¦Ćļ¦ī ņŻ╝ņ×ģ ņĢĮņĀ£ņŚÉ ļö░ļźĖ ņ╣śļŻī ņĀäĒøä ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀź ļ░Å ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗ś ļ│ĆĒÖöļ¤ēņØś ņ░©ņØ┤ļŖö ļ│┤ņØ┤ņ¦Ć ņĢŖņĢśļŗż. ļśÉĒĢ£ ņŻ╝ņ×ģ ņĢĮņĀ£ņŚÉ ļö░ļźĖ ņ╣śļŻīĻ╣īņ¦Ć ņŗ£Ē¢ēļÉ£ ņŻ╝ņé¼ Ēܤņłś ļ░Å ĻĖ░Ļ░äļÅä ņ░©ņØ┤ļź╝ ļ│┤ņØ┤ņ¦Ć ņĢŖņĢśļŗż.
Ļ▓░ļĪĀ
ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Ø ĒÖśņ×ÉņŚÉņä£ ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņ×ģņłĀ ļśÉļŖö ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀ ļ¬©ļæÉ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØś ĻĄ¼ņĪ░ņĀü ļ░Å ĻĖ░ļŖźņĀü ĒÜīļ│ĄņŚÉ ĒÜ©Ļ│╝Ļ░Ć ņ׳ņŚłņ£╝ļéś, ņĢĮņĀ£ņØś ņóģļźśņŚÉ ļö░ļźĖ ĻĖ░ļŖźņĀü ĻĘĖļ”¼Ļ│Ā ĒĢ┤ļČĆĒĢÖņĀü ĒśĖņĀäņØś ņĀĢļÅä, ņ╣śļŻīĻ╣īņ¦Ć ņŗ£Ē¢ēļÉ£ ņŻ╝ņé¼ Ēܤņłś ļ░Å ĻĖ░Ļ░äņØś ņ░©ņØ┤ļŖö ņŚåņŚłļŗż.
ABSTRACT
Purpose
We examined differences in the treatment effects of intravitreal bevacizumab injections and intravitreal aflibercept injections in patients with central serous chorioretinopathy.
Methods
This retrospective analysis included 51 eyes of 49 patients who received intravitreal anti-vascular endothelial growth factor agent injections after initial diagnosis with central serous chorioretinopathy. The patients were divided into two groups: one received an intravitreal bevacizumab injection, and another one received an intravitreal aflibercept injection. Patients with no reaction to treatment or a worsened condition, received repeat treatment with the same therapy. After treatment, patients were monitored for >3 months. Data were collected regarding best- corrected visual acuity (BCVA), subfoveal choroidal thickness, injection number, and treatment duration.
Results
Both intravitreal bevacizumab injections and intravitreal aflibercept injections led to significant differences in BCVA (p < 0.0001, p = 0.001) and subfoveal choroidal thickness (p < 0.0001, p = 0.011), compared between before and after treatment. However, no differences between groups were observed in mean change of BCVA or subfoveal choroidal thickness. In addition, there were no differences between groups in injection number and treatment duration.
Conclusions
In patients with central serous chorioretinopathy, both intravitreal bevacizumab injections and intravitreal aflibercept injections are effective treatment methods. There were no differences between the two medicines in terms of functional and anatomical recovery, or the injection number and treatment duration.
ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Ø(central serous chorioretinopathy)ņØĆ ļ¦źļØĮļ¦ēļ¬©ņäĖĒśłĻ┤Ćņ£╝ļĪ£ļČĆĒä░ ļłäņČ£ļÉ£ ņé╝ņČ£ļ¼╝ļĪ£ ņØĖĒĢ£ ĒÖ®ļ░śļČĆņØś ņŗĀĻ▓Įļ¦Øļ¦ēļ░Ģļ”¼ļź╝ ĒŖ╣ņ¦Ģņ£╝ļĪ£ ĒĢśļŖö ņ¦łļ│æņØ┤ļŗż[1]. ņĀĢĒÖĢĒĢ£ ļ│æņØĖĻ│╝ ļ│æĒā£ņāØļ”¼ļŖö ļ░ØĒśĆņĀĖ ņ׳ņ¦Ć ņĢŖņ£╝ļéś ļ¦źļØĮļ¦ēļ¬©ņäĖĒśłĻ┤ĆņØś ļ╣äņĀĢņāüņĀüņØĖ Ēł¼Ļ│╝ņä▒ ņ”ØĻ░ĆņÖĆ ļ¦Øļ¦ēņāēņåīņāüĒö╝ņØś ĻĖ░ļŖź ņĀĆĒĢśņŚÉ ņØśĒĢ┤ ĒÖ®ļ░śļČĆņØś ļ¦Øļ¦ē ņĢäļל(sub-retina)ņŚÉ ņןņĢĪņä▒ ņĢĪņ▓┤Ļ░Ć ņČĢņĀüļÉśņ¢┤ ļ░£ņāØĒĢśļŖö Ļ▓āņ£╝ļĪ£ ņĢīļĀżņĀĖ ņ׳ļŗż[2]. ĻĖēņä▒ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØś Ļ▓ĮņÜ░ ņØ╝ļ░śņĀüņ£╝ļĪ£ ņ×ÉņŚ░ ĒśĖņĀäļÉśļŖö Ļ▓ĮĻ│╝ļź╝ ļ│┤ņØ┤ņ¦Ćļ¦ī 1ļģä ļé┤ņŚÉ ņ×¼ļ░£ĒĢśļŖö Ļ▓ĮņÜ░Ļ░Ć ļ¦Äļŗż[2]. ņ×äņāüņĀüņ£╝ļĪ£ ņé¼ņÜ®ļÉśļŖö ņ╣śļŻī ļ░®ļ▓Ģņ£╝ļĪ£ļŖö Ļ┤æņŚŁĒĢÖņ╣śļŻīĻ░Ć ņ׳ļŖöļŹ░ ņØ┤ļŖö ļ¦Øļ¦ēņāēņåīņāüĒö╝ņØś ņ£äņČĢ, ļ¦źļØĮļ¦ēļ¬©ņäĖĒśłĻ┤Ć ĒŚłĒśł, ĒÖ®ļ░śņŻ╝ļ│ĆļČĆņØś ņØ┤ņ░© ļ¦źļØĮļ¦ēņŗĀņāØĒśłĻ┤Ć ļ░£ņāØ ļō▒ņØś ĒĢ®ļ│æņ”ØļōżņØ┤ ļ│┤Ļ│ĀļÉśņŚłļŗż[3]. ļĢīļ¼ĖņŚÉ ĻĄ¼ņĪ░ņĀüņØĖ ņåÉņāüņØä ņĄ£ņåīĒÖöĒĢśļ®┤ņä£, ļ¦źļØĮļ¦ēĒśłĻ┤ĆņØś Ļ│╝Ēł¼Ļ│╝ņä▒ņØä ņ£Āļ░£ĒĢśļŖö ĒśłĻ┤Ćļé┤Ēö╝ņä▒ņןņØĖņ×É(vascular endothelial growth factor, VEGF)ļź╝ ņ¢ĄņĀ£ĒĢśļŖö ĒĢŁĒśłĻ┤Ćļé┤Ēö╝ņä▒ņןņØĖņ×É(anti-VEGF agent) ņ╣śļŻīĻ░Ć Ļ│ĀļĀżļÉśņŚłņ£╝ļ®░[4], ņĄ£ĻĘ╝ ņŚ¼ļ¤¼ ņŚ░ĻĄ¼ļōżņŚÉņä£ ņżæņŗ¼ļ¦Øļ¦ēļæÉĻ╗śņØś Ļ░Éņåī, ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀź Ļ░£ņäĀ ļō▒ņØś ĒÜ©Ļ│╝ņĀüņØĖ Ļ▓░Ļ│╝ļź╝ ļ│┤ņśĆļŗż[5-11].
Anti-VEGF agentņŚÉņä£ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö(AvastinŌōć, Genentech, South San Francisco, CA, USA)ņØĆ VEGF-Aļź╝ ņ¢ĄņĀ£ĒĢśņŚ¼ ņ×æņÜ®ĒĢśņ¦Ćļ¦ī[12,13], ņØ┤Ēøä Ļ░£ļ░£ļÉ£ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ(EyleaŌōć, Regeneron Pharmaceuticals, Inc, Tarrytown, NY, USA; Bayer Pharma AG, Berlin, Germany)ļŖö VEGF-Aļ┐É ņĢäļŗī VEGF-BņÖĆ Ēā£ļ░śņä▒ņןņØĖņ×É(placental growth factor)ņŚÉļÅä Ļ▓░ĒĢ®ĒĢśņŚ¼ ņ×æņÜ®ĒĢśļŖö ĒŖ╣ņ¦Ģ[14]Ļ│╝ VEGF-AņŚÉ ļŹö ļ╣Āļź┤Ļ▓ī Ļ▓░ĒĢ®ĒĢśļ®░ 100ļ░░ ļåÆņØĆ Ļ▓░ĒĢ®ļĀźņØä ņ¦Ćļŗī ņןņĀÉņØä Ļ░ĆņĪīļŗż[15-18]. ĻĘĖļ¤¼ļéś ĻĄŁļé┤ņŚÉņä£ļŖö ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØ┤ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖņØś ļ│┤ĒŚśņĀüņÜ® ļīĆņāü ņ¦łĒÖśņØ┤ ņĢäļŗłĻĖ░ņŚÉ, ņé¼ņÜ® ņŗ£ ĒÖśņ×ÉĻ░Ć ņĢĮĻ░ÆņØä ņĀäļČĆ ļČĆļŗ┤ĒĢ┤ņĢ╝ ĒĢśļŖö Ļ▓ĮņĀ£ņĀüņØĖ ņÜöņØĖņ£╝ļĪ£ ņØĖĒĢśņŚ¼ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Ø ĒÖśņ×ÉņŚÉņä£ ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀņØä ņŗ£Ē¢ēĒĢśĻĖ░ ņē¼ņÜ┤ ņŚ¼Ļ▒┤ņØ┤ ņĢäļŗłļŗż. ļśÉĒĢ£ ĻĄŁļé┤ņÖĖņŚÉņä£ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņŚÉņä£ ļæÉ ņĢĮņĀ£ļź╝ ļ╣äĻĄÉĒĢ£ ņŚ░ĻĄ¼ļŖö ļō£ļ¼╝ļŗż. ņØ┤ņŚÉ ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ļŖö ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Ø ĒÖśņ×ÉņŚÉņä£ ņĢĮņĀ£ ņóģļźśņŚÉ ļö░ļźĖ ņŗ£ļĀź ļ░Å ņżæņŗ¼ļ¦źļØĮļ¦ē ļæÉĻ╗śņØś ļ│ĆĒÖö, ņ╣śļŻīņŚÉ ĒĢäņÜöĒĢ£ ĻĖ░Ļ░ä ļ░Å ņŻ╝ņé¼ Ēܤņłś ļō▒ņØä ļČäņäØĒĢ©ņ£╝ļĪ£ņŹ©, ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņ×ģņłĀĻ│╝ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀņØś ĒÜ©Ļ│╝ļź╝ ļ╣äĻĄÉĒĢ┤ ļ│┤Ļ│Āņ×É ĒĢśņśĆļŗż.
2010ļģä 1ņøöļČĆĒä░ 2020ļģä 7ņøöĻ╣īņ¦Ć ļ│ĖņøÉņŚÉņä£ ņ▓śņØī ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Øņ£╝ļĪ£ ņ¦äļŗ©ļ░øĻ│Ā ņĄ£ņåī 3Ļ░£ņøö ļÅÖņĢł ņŗ£ļĀźĻ│╝ ĒÖ®ļ░śļČĆņØś ņĢłņĀĢļÉ£ ņåīĻ▓¼ņØä ļ│┤ņØ╝ ļĢīĻ╣īņ¦Ć ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖņŻ╝ņ×ģņłĀ Ēś╣ņØĆ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀņØä ņŗ£Ē¢ē ļ░øņØĆ ņ╣śļŻīĻĄ░(treatment group) ņ┤Ø 49ļ¬ģ 51ņĢłņØś ņØśļ¼┤ĻĖ░ļĪØņØä ĒøäĒ¢źņĀüņ£╝ļĪ£ ļČäņäØĒĢśņśĆļŗż. ļŗżņØī ĻĖ░ņżĆņØä ņČ®ņĪ▒ĒĢśļŖö Ļ▓ĮņÜ░ ņŚ░ĻĄ¼ņŚÉ ĒżĒĢ©ĒĢśņśĆļŗż: 1) ņŖżĒÄÖĒŖĖļ¤╝ ļÅäļ®öņØĖ ļ╣øĻ░äņäŁļŗ©ņĖĄņ┤¼ņśü(Spectralis OCTŌōć, Heidelberg Engineering, Heidelberg, Germany)ņŚÉņä£ ļ¬ģĒÖĢĒĢśĻ▓ī ļ¦Øļ¦ēĒĢśņĢĪ(subretinal fluid, SRF)ņØ┤ Ļ┤Ćņ░░ļÉśļŖö Ļ▓ĮņÜ░, 2) ĒśĢĻ┤æņĢłņĀĆĒśłĻ┤ĆņĪ░ņśüņłĀ(Spectralis HRA2Ōōć, Heidelberg Engineering, Heidelberg, Germany)ņŚÉņä£ ļ¦Øļ¦ēĒĢśņĢĪĻ│╝ ņŚ░Ļ┤ĆļÉ£ ļłäņČ£ņĀÉņØ┤ Ļ┤Ćņ░░ļÉśļŖö Ļ▓ĮņÜ░, 3) ļ¦łņ¦Ćļ¦ē ņĢłĻĄ¼ļé┤ņŻ╝ņé¼ Ēøä 3Ļ░£ņøö ņØ┤ņāüņØś Ļ▓ĮĻ│╝ Ļ┤Ćņ░░ņØ┤ Ļ░ĆļŖźĒĢ£ Ļ▓ĮņÜ░. ļŗżņØīĻ│╝ Ļ░ÖņØĆ Ļ▓ĮņÜ░ļŖö ņŚ░ĻĄ¼ņŚÉņä£ ņĀ£ņÖĖĒĢśņśĆļŗż: 1) ļ¦źļØĮļ¦ēņŗĀņāØĒśłĻ┤Ć(choroidal neovascularization, CNV), ņŚ░ļĀ╣Ļ┤ĆļĀ©ĒÖśļ░śļ│Ćņä▒ ļō▒ ļŗżļźĖ ĒÖ®ļ░śņ¦łĒÖśņØ┤ ņ׳ļŖö Ļ▓ĮņÜ░, 2) ņŻ╝ņé¼ ņ╣śļŻī ĻĖ░Ļ░ä ņżæ ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ĒŖĖļ”¼ņĢöņŗ£ļåĆļĪĀ ņŻ╝ņ×ģņłĀ, Ļ┤æņŚŁĒĢÖņ╣śļŻī, ļ¦Øļ¦ēĻ┤æņØæĻ│ĀņłĀ ļō▒ ļŗżļźĖ ņŗ£ņłĀņØä ļ░øņØĆ Ļ▓ĮņÜ░, 3) ņŻ╝ņé¼ņ╣śļŻī ĻĖ░Ļ░ä ņżæ ņ£Āļ”¼ņ▓┤ņĀłņĀ£ņłĀ, ļ░▒ļé┤ņןņłśņłĀ ļō▒ ļŗżļźĖ ņłśņłĀņØä ĒĢ£ Ļ▓ĮņÜ░, 4) ļÅÖņØ╝ ņĢĮņĀ£ļź╝ ņé¼ņÜ®ĒĢśņ¦Ć ņĢŖĻ│Ā ņżæĻ░äņŚÉ ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖņŚÉņä£ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ Ēś╣ņØĆ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖņŚÉņä£ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö Ļ░ÖņØ┤ ņĢĮņĀ£Ļ░Ć ļ░öļĆÉ Ļ▓ĮņÜ░. ļ│Ė ņŚ░ĻĄ¼ļŖö ĒŚ¼ņŗ▒ĒéżņäĀņ¢ĖņØä ņżĆņłśĒĢśņśĆņ£╝ļ®░, ļ│ĖņøÉ ņ×äņāüņŚ░ĻĄ¼ņ£żļ”¼ņŗ¼ņØśņ£äņøÉĒÜī(Institutional Review Board, IRB)ņØś ņŖ╣ņØĖ ĒĢśņŚÉ ņ¦äĒ¢ēļÉśņŚłļŗż(IRB ņŖ╣ņØĖ ļ▓łĒśĖ: 2021-01-006-001).
ĒÖśņ×ÉĻ░Ć ņ▓śņØī ņÖĖļלļź╝ ļé┤ņøÉĒĢśņśĆņØä ļĢī ņŖżļä¼ļĀīņŗ£ļĀźĒæ£ļź╝ ņØ┤ņÜ®ĒĢ£ ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀź ņĖĪņĀĢ, ļ╣äņĀæņ┤ēņĢłņĢĢĻ│ä(KT-800Ōōć, Kowa American Corp., New York, NY, USA)ļź╝ ņØ┤ņÜ®ĒĢ£ ņĢłņĢĢ ņĖĪņĀĢ, ņĢłņĀĆĻ▓Ćņé¼, ņŖżĒÄÖĒŖĖļ¤╝ ļÅäļ®öņØĖ ļ╣øĻ░äņäŁļŗ©ņĖĄņ┤¼ņśü, ĒśĢĻ┤æņĢłņĀĆĒśłĻ┤ĆņĪ░ņśüņłĀņØ┤ ņŗ£Ē¢ēļÉśņŚłļŗż. ņ¦äļŗ© Ēøä 1ņŻ╝ņØ╝ ņØ┤ļé┤ņŚÉ ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņ×ģņłĀ Ēś╣ņØĆ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀņØä ņŗ£Ē¢ēĒĢśņśĆļŗż. ņĢłĻĄ¼ ļé┤ ņŻ╝ņé¼ ņĀäņŚÉ ļ¬©ļōĀ ĒÖśņ×ÉņŚÉĻ▓ī Ļ▓ĮĻ│╝ Ļ┤Ćņ░░, Ļ┤æņŚŁĒĢÖņ╣śļŻī ļō▒ ļŗżļźĖ ņ╣śļŻī ļ░®ļ▓ĢļōżņØä ņäżļ¬ģĒĢśņśĆĻ│Ā, ņŻ╝ņé¼ ņĢĮņĀ£ļōżņŚÉ ļīĆĒĢ£ ņČ®ļČäĒĢ£ ņäżļ¬ģĻ│╝ ĒĢ©Ļ╗ś ĒÖśņ×ÉĻ░Ć ņ¦üņĀæ ņĢĮņĀ£ļź╝ ņäĀĒāØĒĢśļÅäļĪØ ĒĢśņśĆņ£╝ļ®░, ļśÉĒĢ£ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņŚÉņä£ ĒŚłĻ░Ćņ┤łĻ│╝ņÜ®ņĀ£ļĪ£ ņé¼ņÜ®ļÉśļŖö ņĢĮņĀ£ņ×äņØä ņäżļ¬ģĒĢśĻ│Ā ņé¼ņĀä ļÅÖņØśļź╝ ļ░øņĢśļŗż.
ņŻ╝ņé¼ Ēøä 1ņØ╝ņ¦Ė ņÖĖļלļź╝ ļ░®ļ¼ĖĒĢśņŚ¼ ņĢłņĢĢņāüņŖ╣ ņŚ¼ļČĆ ļ░Å ņĢłļé┤ ņŚ╝ņ”Ø ļ░£ņāØ ņ£Āļ¼┤ ļō▒ņØś ĒĢ®ļ│æņ”Ø ņŚ¼ļČĆļź╝ ĒÖĢņØĖĒĢśņśĆņ£╝ļ®░, ņĢĮ 1Ļ░£ņøö Ļ░äĻ▓®ņ£╝ļĪ£ ņČöņĀü Ļ┤Ćņ░░ņØä ņŗ£Ē¢ēĒĢśņśĆļŗż. ļ¦ż ĒÜīņØś ņČöņĀü Ļ┤Ćņ░░ņŚÉņä£ ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀźĻ│╝ ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗ś ņĖĪņĀĢņØ┤ ņØ┤ļŻ©ņ¢┤ņĪīļŗż. ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ ŌĆśņ╣śļŻīŌĆÖļŖö 3Ļ░£ņøö ņØ┤ņāüņØś Ļ▓ĮĻ│╝ Ļ┤Ćņ░░ņŚÉņä£ ļ╣øĻ░äņäŁļŗ©ņĖĄņ┤¼ņśüņāü SRFĻ░Ć ņé¼ļØ╝ņ¦ä Ļ▓āņ£╝ļĪ£ ņĀĢņØśĒĢśņśĆļŗż. ņČöņĀü Ļ┤Ćņ░░ ņżæ ļ╣øĻ░äņäŁļŗ©ņĖĄņ┤¼ņśüņŚÉņä£ ļ¦Øļ¦ēĒĢśņĢĪņØś ņ¦ĆņåŹ Ēś╣ņØĆ ņ”ØĻ░ĆĻ░Ć ĒÖĢņØĖļÉśļŖö Ļ▓ĮņÜ░ ņŻ╝ņé¼ņ╣śļŻīņŚÉ ļ░śņØæņØ┤ ņŚåĻ▒░ļéś ņĢģĒÖöĻ░Ć ļÉśļŖö Ļ▓āņ£╝ļĪ£ Ļ░äņŻ╝ĒĢśņśĆņ£╝ļ®░ ņČöĻ░Ć ņŻ╝ņé¼ņ╣śļŻīĻ░Ć ņŗ£Ē¢ēļÉśņŚłļŗż.
ĒåĄĻ│äņĀü ļČäņäØņØä ņ£äĒĢ┤ ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀźņØĆ logarithm of the minimal angle of resolution (logMAR) scaleļĪ£ ļ│ĆĒÖśĒĢśņśĆļŗż. ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗śļŖö ļ╣øĻ░äņäŁļŗ©ņĖĄņ┤¼ņśü ĻĖ░ĻĖ░ņØś ļööņ¦ĆĒäĖ ņ║śļ”¼ĒŹ╝ĻĖ░ļŖźņØä ņé¼ņÜ®ĒĢśņŚ¼ ņżæņŗ¼ņÖĆ ņ£äņ╣śņŚÉņä£ ļ¦Øļ¦ēņāēņåīņāüĒö╝ļĪ£ļČĆĒä░ ļ¦źļØĮļ¦ē-Ļ│Ąļ¦ē ņĢłņ¬Į Ļ▓ĮĻ│äĻ╣īņ¦ĆņØś Ļ▒░ļ”¼ļź╝ ņłśņ¦üņ£╝ļĪ£ ņĖĪņĀĢĒĢśņśĆļŗż. ĒÖśņ×ÉļŖö ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņ×ģņłĀņØä ņŗ£Ē¢ē ļ░øņØĆ ĻĄ░(ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ĻĄ░)Ļ│╝ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀņØä ņŗ£Ē¢ē ļ░øņØĆ ĻĄ░(ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░) ļæÉ ĻĄ░ņ£╝ļĪ£ ļéśļłäņŚłļŗż. ņ╣śļŻī ņĀäĻ│╝ ņ╣śļŻī Ēøä 3Ļ░£ņøöņ¦ĖņØś ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀź, ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗śļź╝ ļ╣äĻĄÉĒĢśņśĆņ£╝ļ®░, ļæÉ ĻĄ░ Ļ░ä ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀźĻ│╝ ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗śņØś ļ│ĆĒÖöļ¤ē ļ░Å ņŗ£Ē¢ēļÉ£ ņŻ╝ņé¼ Ēܤņłś, ņ╣śļŻīĻ╣īņ¦ĆņØś ĻĖ░Ļ░äņØä ļ╣äĻĄÉĒĢśņśĆļŗż.
ļŹ░ņØ┤Ēä░ļČäņäØņŚÉļŖö SPSS Statistics 18 (IBM, Armonk, NY, USA) ņåīĒöäĒŖĖņø©ņ¢┤ļź╝ ņØ┤ņÜ®ĒĢśņśĆļŗż. ļ│Ė ņŚ░ĻĄ¼ļŖö Ēæ£ļ│ĖņłśĻ░Ć ņĀüņ¢┤ ļ¬©ņ¦æļŗ©ņØś ņĀĢĻĘ£ņä▒ Ļ▓ĆĒåĀļź╝ ņ£äĒĢśņŚ¼ Kolmogorov-SmirnovņØś ņĀĢĻĘ£ņä▒ Ļ▓ĆņĀĢņØä ņŗżņŗ£ĒĢśņśĆļŗż. ņĀĢĻĘ£ņä▒ņØä ļ│┤ņØ┤ļŖö ļæÉ ĻĄ░ Ļ░äņØś ļ╣äĻĄÉļŖö independent t-testļź╝, ņĀĢĻĘ£ņä▒ņØä ļ│┤ņØ┤ņ¦Ć ņĢŖļŖö ļæÉ ĻĄ░ņØĆ Mann-Whitney testļź╝ ĒåĄĒĢśņŚ¼ Ļ▓ĆņĀĢĒĢśņśĆļŗż. ņĀĢĻĘ£ņä▒ņØä ļ│┤ņØ┤ļŖö ņĀäĒøä ļ╣äĻĄÉļŖö paired t-testļź╝, ņĀĢĻĘ£ņä▒ņØä ļ│┤ņØ┤ņ¦Ć ņĢŖļŖö ņĀäĒøä ļ╣äĻĄÉļŖö Wilcoxon signed rank testļź╝ ņé¼ņÜ®ĒĢśņśĆļŗż. ļ¬ģļ¬®ņ▓ÖļÅäņØś ņ×ÉļŻī ļČäņäØņŚÉļŖö chi-squared testļź╝ ĒåĄĒĢśņŚ¼ Ļ▓ĆņĀĢĒĢśņśĆļŗż. ļ¬©ļōĀ Ļ▓░Ļ│╝Ļ░ÆņØĆ pĻ░ÆņØ┤ 0.05 ļ»Ėļ¦īņØ╝ Ļ▓ĮņÜ░ ĒåĄĻ│äņĀüņ£╝ļĪ£ ņ£ĀņØśĒĢ£ Ļ▓āņ£╝ļĪ£ Ļ░äņŻ╝ĒĢśņśĆļŗż.
ņĀäņ▓┤ 51ņĢł(49ļ¬ģ) ņżæ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ĻĄ░ņØ┤ 32ņĢł, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░ņØ┤ 19ņĢłņØ┤ņŚłļŗż. ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖĻĄ░ņŚÉņä£ļŖö ļé©ņ×É 18ņĢł(56.3%)ņ£╝ļĪ£ ĒÅēĻĘĀņŚ░ļĀ╣ņØĆ 50.44 ┬▒ 7.14ņäĖņśĆĻ│Ā, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖĻĄ░ņŚÉņä£ļŖö ļé©ņ×É 15ņĢł(78.9%)ņ£╝ļĪ£ ĒÅēĻĘĀņŚ░ļĀ╣ņØĆ 55.79 ┬▒ 12.39ņäĖņśĆļŗż. ņ╣śļŻī ņĀä ĒÖśņ×ÉĻĄ░ņØś ĒŖ╣ņä▒ņØĆ Table 1ņŚÉ ļéśĒāĆļéś ņ׳ļŗż.
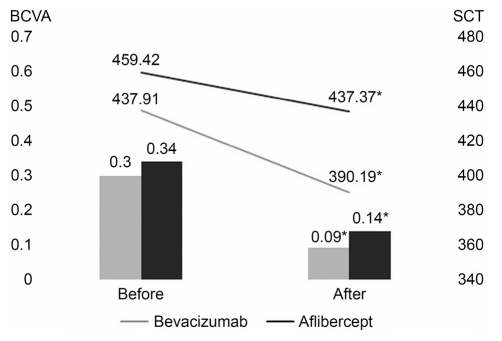
ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ĻĄ░ņØś ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀźņØĆ ņ╣śļŻī ņĀä 0.30 ┬▒ 0.22ņŚÉņä£ ņ╣śļŻī Ēøä 0.09 ┬▒ 0.10ņ£╝ļĪ£, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░ņØĆ ņ╣śļŻī ņĀä 0.34 ┬▒ 0.23ņŚÉņä£ ņ╣śļŻī Ēøä 0.14 ┬▒ 0.16ņ£╝ļĪ£ ĒåĄĻ│äņĀüņ£╝ļĪ£ ņØśļ»Ė ņ׳Ļ▓ī ņóŗņĢäņĪīņ£╝ļ®░(Ļ░üĻ░ü, p<0.0001, p=0.001), ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗ś(╬╝m) ļśÉĒĢ£ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ĻĄ░ņŚÉņä£ ņ╣śļŻī ņĀä 437.9 1 ┬▒ 79.72ņŚÉņä£ ņ╣śļŻī Ēøä 390.19 ┬▒ 74.72ļĪ£, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░ņØĆ ņ╣śļŻī ņĀä 459.42 ┬▒ 45.14ņŚÉņä£ ņ╣śļŻī Ēøä 437.37 ┬▒ 37.38ļĪ£ ĒåĄĻ│äņĀüņ£╝ļĪ£ ņØśļ»Ė ņ׳Ļ▓ī Ļ░ÉņåīĒĢśņśĆļŗż(Ļ░üĻ░ü, p<0.0001, p=0.011) (Fig. 1).
ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖņÖĆ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ļæÉ ĻĄ░ Ļ░ä ņ╣śļŻī ņĀäĒøä ļČäņäØņŚÉņä£ ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀźņØś ļ│ĆĒÖöļ¤ēņØĆ ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖĻĄ░ņŚÉņä£ 0.21 ┬▒ 0.21, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░ņŚÉņä£ 0.20 ┬▒ 0.17ņØ┤ņŚłĻ│Ā, ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗ś ļ│ĆĒÖöļ¤ēņØĆ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ĻĄ░ņŚÉņä£ 47.72 ┬▒ 57.78, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░ņŚÉņä£ 22.05 ┬▒ 30.36ņØ┤ņŚłņ£╝ļéś ĒåĄĻ│äņĀüņ£╝ļĪ£ ņ£ĀņØśĒĢ£ ņ░©ņØ┤ļŖö ņŚåņ¢┤, ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņ×ģņłĀĻ│╝ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀ Ļ░äņØś ĻĖ░ļŖźĻ│╝ ĒĢ┤ļČĆĒĢÖņĀü ĒÜīļ│ĄņØś ņ░©ņØ┤ļź╝ ļ│┤ņØ┤ņ¦Ć ņĢŖņĢśļŗż(Table 2).
ņ╣śļŻīĻ╣īņ¦Ć Ļ▒Ėļ”░ ĻĖ░Ļ░äņØĆ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ĻĄ░ņØ┤ 101.97 ┬▒ 100.42ņØ╝, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░ņØ┤ 80.74 ┬▒ 75.80ņØ╝ņØ┤ Ļ▒ĖļĀĖņ£╝ļéś ĒåĄĻ│äņĀüņ£╝ļĪ£ ņ£ĀņØśĒĢ£ ņ░©ņØ┤ļŖö ņŚåņŚłļŗż(p=0.453). 1ĒÜī ņŻ╝ņé¼ņ╣śļŻī ņØ┤Ēøä ĒśĖņĀäļÉśņ¦Ć ņĢŖņĢä ņČöĻ░Ć ņ╣śļŻīļź╝ ĒĢ£ Ļ▓ĮņÜ░ļŖö ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ĻĄ░ņŚÉņä£ 32ņĢł ņżæ ņ┤Ø 7ņĢłņØ┤ņŚłņ£╝ļ®░, ĻĘĖ ņżæ 3ņĢł(9.38%)ņØ┤ 2ĒÜī, 4ņĢł(12.5%)ņØ┤ 3ĒÜī ņŻ╝ņé¼ļź╝ ņŗ£Ē¢ēĒĢśņśĆļŗż. ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░ņØĆ 19ņĢł ņżæ ņ┤Ø 6ņĢłņØ┤ ņČöĻ░Ć ņ╣śļŻīļź╝ ņŗ£Ē¢ē ļ░øņĢśņ£╝ļ®░, ĻĘĖ ņżæ 4ņĢł(21.05%)ņØ┤ 2ĒÜī, 1ņĢł(5.26%)ņØ┤ 3ĒÜī, 1ņĢł(5.26%)ņØ┤ 4ĒÜī ņŻ╝ņé¼ļź╝ ņŗ£Ē¢ē ļ░øņĢśļŗż. Ļ▓░Ļ│╝ņĀüņ£╝ļĪ£ ņ╣śļŻīĻ╣īņ¦ĆņØś ņŻ╝ņé¼ ĒܤņłśļŖö ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ĻĄ░ņØ┤ 1.34 ┬▒ 0.70ĒÜī, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░ņØ┤ 1.47 ┬▒ 0.84ĒÜīļĪ£ ĒåĄĻ│äņĀüņ£╝ļĪ£ ņ£ĀņØśĒĢ£ ņ░©ņØ┤ļŖö ņŚåņŚłļŗż(p=0.499). ņĀäņ▓┤ Ļ▓ĮĻ│╝ Ļ┤Ćņ░░ ĻĖ░Ļ░ä ļÅÖņĢł 51ņĢłņŚÉ ļīĆĒĢśņŚ¼ ņĢłņĢĢņāüņŖ╣, ņĢłļé┤ņŚ╝, ļ¦Øļ¦ēļ░Ģļ”¼, ņ£Āļ”¼ņ▓┤ņČ£Ēśł ļ░Å ņĀäņŗĀņĀü ļČĆņ×æņÜ®ņØĆ Ļ┤Ćņ░░ļÉśņ¦Ć ņĢŖņĢśļŗż.
ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØś ļ│æļ”¼ĻĖ░ņĀäņØĆ ņśüņāüĻ▓Ćņé¼ĻĖ░ņłĀņØś ļ░£ņĀäĻ│╝ ņłśļ¦ÄņØĆ ņŚ░ĻĄ¼ļōżņŚÉļÅä ļČłĻĄ¼ĒĢśĻ│Ā ņŚ¼ļ¤¼ Ļ░Ćņ¦Ć Ļ░ĆņäżļōżņØ┤ ņĀ£ņŗ£ļÉśņŚłņØä ļ┐É ļ¬ģĒÖĢĒĢśĻ▓ī ļ░ØĒśĆņ¦Ćņ¦Ć ņĢŖņĢśļŗż. ņĄ£ĻĘ╝ Ļ░Ćņן ņ£ĀļĀźĒĢ£ Ļ░ĆņäżņØĆ ļ¦źļØĮļ¦ēļ¬©ņäĖĒśłĻ┤ĆņØś Ēł¼Ļ│╝ņä▒ ņ”ØĻ░ĆņØ┤ļŗż[19]. ļ¦źļØĮļ¦ēņØś ņÜĖĒśł, ĒŚłĒśł ļśÉļŖö ņŚ╝ņ”Ø ļō▒ņŚÉ ņØśĒĢ£ ļ¦źļØĮļ¦ēļ¬©ņäĖĒśłĻ┤ĆņØś Ļ│╝Ēł¼Ļ│╝ņä▒ ļĢīļ¼ĖņŚÉ ļ¦źļØĮļ¦ē ļé┤ ņĀĢņłśņĢĢņØ┤ ņ”ØĻ░ĆĒĢśĻ│Ā[20], ļ¦Øļ¦ēņāēņåīņāüĒö╝ļ░Ģļ”¼Ļ░Ć ņ┤ēņ¦äļÉśņ¢┤ Ļ░ÉĻ░üņŗĀĻ▓Įļ¦Øļ¦ēĻ│╝ ļ¦Øļ¦ēņāēņåīņāüĒö╝ ņé¼ņØ┤ņŚÉ ņé╝ņČ£ļ¼╝ņØ┤ ņČĢņĀüļÉ£ļŗżļŖö Ļ░ĆņäżņØ┤ļŗż. ĻĖēņä▒ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØĆ 1-4Ļ░£ņøö ņĢłņŚÉ Ļ░ÉĻ░üņŗĀĻ▓Įļ¦Øļ¦ēņØś ņ×¼ļČĆņ░®Ļ│╝ ĒĢ©Ļ╗ś ņŗ£ļĀźņØ┤ ĒÜīļ│ĄļÉśļŖö ņ×ÉņŚ░ ĒśĖņĀäņØś Ļ▓ĮĻ│╝ļź╝ ļ│┤ņØ┤ĻĖ░ļÅä ĒĢ£ļŗż[19]. ĻĘĖļ¤¼ļéś 30-50% ĒÖśņ×ÉņŚÉņä£ 1ļģä ņĢłņŚÉ ņ×¼ļ░£ņØ┤ ĒØöĒĢśļ®░, ļ╣łļ▓łĒĢ£ ņ×¼ļ░£ņØĆ ļ¦Øļ¦ēņāēņåīņāüĒö╝ņØś ņ£äņČĢĻ│╝ Ļ░ÉĻ░üņŗĀĻ▓Įļ¦Øļ¦ēņØś ļ│ĆĒÖöļź╝ ņØ╝ņ£╝ņ╝£ ņŗ£Ļ░üĻĖ░ļŖźņØä ņśüĻĄ¼Ē׳ ņ×āņØä ņłś ņ׳ĻĖ░ ļĢīļ¼ĖņŚÉ Ļ┤æņŚŁĒĢÖņ╣śļŻī ļ░Å ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ anti-VEGF ņŻ╝ņ×ģņłĀ Ļ░ÖņØĆ ņ╣śļŻīļź╝ ņŗ£ļÅäĒĢ┤ņÖöļŗż.
Anti-VEGF ņ╣śļŻīļŖö ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņŚÉņä£ ļ¦źļØĮļ¦ēĻ│╝ ļ¦Øļ¦ēņāēņåīņāüĒö╝ņØś ĻĖ░ļŖź ļ░Å ĻĄ¼ņĪ░ ļ│ĆĒÖöļĪ£ ņØĖĒĢ┤ ņĀĆĻ┤Ćļźś, ņĀĆņé░ņåīņāüĒā£Ļ░Ć ņ┤łļלļÉśļ®┤ ĒśłĻ┤Ćļé┤Ēö╝ņä▒ņןņØĖņ×É(VEGF)Ļ░Ć ļ░£ĒśäļÉ£ļŗżļŖö Ļ░Ćņäż ĒĢśņŚÉ ņé¼ņÜ®ļÉśņŚłņ£╝ļ®░[21], ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ņŚÉ anti-VEGFļź╝ ņŻ╝ņé¼ĒĢśņŚ¼ VEGFļź╝ ņ¢ĄņĀ£ņŗ£Ēéżļ®┤ ļ¦źļØĮļ¦ēĒśłĻ┤ĆņØś Ļ│╝Ēł¼Ļ│╝ņä▒ņŚÉ ļ│ĆĒÖöļź╝ ņŻ╝ņ¢┤ ļ¦Øļ¦ēĒĢśņĢĪņØä Ļ░Éņåīņŗ£Ēé¼ ņłś ņ׳ļŗżļŖö ļ│┤Ļ│ĀĻ░Ć ņ׳ļŗż.16,17 ĻĖēņä▒ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņŚÉņä£ ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖņØ┤ļéś ļØ╝ļŗłļ╣äņŻ╝ļ¦Ö(LucentisŌōć, Genentech Inc., South San Francisco, CA, USA) ņ╣śļŻīĻĄ░ņØ┤ Ļ▓ĮĻ│╝ Ļ┤Ćņ░░ĻĄ░ļ│┤ļŗż ĒĢ┤ļČĆĒĢÖņĀü ĒśĖņĀäņØ┤ ļ╣Āļź┤ļŗżļŖö ņŚ░ĻĄ¼ Ļ▓░Ļ│╝ļōżņØ┤ ņ׳ņ£╝ļ®░[22-24], Koh et al [25]ņØĆ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņ╣śļŻīĻ░Ć ĻĖēņä▒ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØś ņ×¼ļ░£ļźĀņØä ļé«ņČöļŖö ļŹ░ņŚÉ ĻĖ░ņŚ¼ĒĢ£ļŗżĻ│Ā ĒĢśņśĆļŗż. Jung et al [26]ņØĆ ĻĖēņä▒ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņŚÉņä£ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņ╣śļŻīĻĄ░ņØ┤ ņØśļ»Ė ņ׳ļŖö ņżæņŗ¼ļ¦Øļ¦ēļæÉĻ╗śņÖĆ ļ¦źļØĮļ¦ēļæÉĻ╗śņØś ĒśĖņĀäņØä ļ│┤ņśĆĻ│Ā, ļīĆņĪ░ĻĄ░ļ│┤ļŗż ļŹö ļ╣ĀļźĖ ņŗ£ļĀź ĒśĖņĀäņØä ļ│┤ņśĆļŗżĻ│Ā ļ│┤Ļ│ĀĒĢśņśĆļŗż. ļ¦īņä▒ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØä ļīĆņāüņ£╝ļĪ£ ĒĢ£ ņĀäĒ¢źņĀüņØĖ ņŚ░ĻĄ¼ņŚÉņä£ ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖĻ│╝ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņ╣śļŻīĻ░Ć ĒĢ┤ļČĆĒĢÖņĀü ļ░Å ĻĖ░ļŖź ĒśĖņĀäņØä ļ│┤ņØĖļŗżļŖö ņŚ░ĻĄ¼ Ļ▓░Ļ│╝ļōżļÅä ņ׳ļŗż[27,28]. ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ļÅä ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ anti-VEGF ņŻ╝ņé¼ļź╝ ņ┤łĻĖ░ ņ╣śļŻīļĪ£ ņé¼ņÜ®ĒĢ£ ĒÖśņ×ÉļōżņØä ļīĆņāüņ£╝ļĪ£ ņŚ░ĻĄ¼ļź╝ ņ¦äĒ¢ēĒĢśņŚ¼ ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖĻĄ░Ļ│╝ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖĻĄ░ ļ¬©ļæÉ ĒĢ®ļ│æņ”Ø ņŚåņØ┤ ņĄ£ļīĆ ĻĄÉņĀĢņŗ£ļĀźņØś ĒśĖņĀäĻ│╝ ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗ś Ļ░ÉņåīļØ╝ļŖö ĻĖ░ļŖźņĀü, ĒĢ┤ļČĆĒĢÖņĀüņ£╝ļĪ£ ņ£ĀņØśĒĢ£ ĒÜ©Ļ│╝ļź╝ Ļ┤Ćņ░░ĒĢĀ ņłś ņ׳ņŚłļŗż.
ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Ø ņÖĖņŚÉ ļīĆĒæ£ņĀüņØĖ ļ¦Øļ¦ēĒśłĻ┤Ćņ¦łĒÖśņØĖ ļŗ╣ļć©ļ¦Øļ¦ēļ│æņ”ØņØ┤ļéś ņżæņŗ¼ļ¦Øļ¦ēņĀĢļ¦źĒÅÉņćäņŚÉņä£ļŖö ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖĻ░Ć ļ▓Āļ░öņŗ£ņŻ╝ļ¦Öļ│┤ļŗż ĒÜ©Ļ│╝ņĀüņØ┤ļØ╝ļŖö ņŚ░ĻĄ¼ Ļ▓░Ļ│╝ļōżņØ┤ ņ׳ļŗż. Wells et al [29]Ļ│╝ Heier et al [30]ļŖö ļŗ╣ļć©ļ¦Øļ¦ēļ│æņ”ØņŚÉņä£ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖĻ░Ć ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖņØä ņé¼ņÜ®ĒĢ£ ĻĄ░ļ│┤ļŗż ņŗ£ļĀźĒśĖņĀäņŚÉ ņ׳ņ¢┤ ĒÜ©Ļ│╝ņĀüņØ┤ļØ╝Ļ│Ā ĒĢśņśĆļŗż. Lotfy et al [31]ņØĆ ņżæņŗ¼ļ¦Øļ¦ēņĀĢļ¦źĒÅÉņćäņŚÉņä£ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖņÖĆ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ļ¬©ļæÉ ĒÜ©Ļ│╝Ļ░Ć ņ׳ņŚłņ¦Ćļ¦ī, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖļź╝ ņé¼ņÜ®ĒĢ£ ĻĄ░ņŚÉņä£ ļŹö ĻĖ┤ ņŻ╝ņé¼ Ļ░äĻ▓®Ļ│╝ ļŹö ņĀüņØĆ ĒܤņłśņØś ņŻ╝ņé¼ļĪ£ ĒÜ©Ļ│╝Ļ░Ć ļéśĒāĆļé¼ĻĖ░ ļĢīļ¼ĖņŚÉ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖĻ░Ć ļ▓Āļ░öņŗ£ņŻ╝ļ¦Öļ│┤ļŗż ĒÜ©Ļ│╝ņĀüņØ┤ļ®░, ņØ┤ļŖö ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖņØś ņäĖĒżņłśņÜ®ņ▓┤ Ļ▓░ĒĢ® ļŖźļĀźņØ┤ ļŹö ļåÆņĢä VEGFļź╝ ņ¢ĄņĀ£ĒĢśļŖö ļŖźļĀźņØ┤ ļŹö ņóŗĻĖ░ ļĢīļ¼ĖņØ┤ļØ╝Ļ│Ā ĒĢśņśĆļŗż. ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņŚÉņä£ ļæÉ ņĢĮņĀ£ļź╝ ļ╣äĻĄÉĒĢ£ ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ļŖö ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ĻĄ░Ļ│╝ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ĻĄ░ Ļ░äņØś ņ╣śļŻī ņĀä Ēøä ņĄ£ļīĆĻĄÉņĀĢņŗ£ļĀź ļ░Å ņżæņŗ¼ļ¦źļØĮļ¦ēļæÉĻ╗ś ļ│ĆĒÖöļ¤ēņØä ļ╣äĻĄÉĒĢśņśĆņØä ļĢī ņ£ĀņØśĒĢ£ ņ░©ņØ┤ļź╝ ļ│┤ņØ┤ņ¦Ć ņĢŖņĢśļŗż. ņØ┤ļŖö ņĢ×ņä£ ņ¢ĖĻĖēĒĢ£ ļŗżļźĖ ļ¦Øļ¦ēĒśłĻ┤Ćņ¦łĒÖśļōżņØś ņŚ░ĻĄ¼ņÖĆļŖö ņāüņØ┤ĒĢ£ Ļ▓░Ļ│╝ņØ┤ļŗż. ļ│Ė ņŚ░ĻĄ¼ņÖĆ ļ╣äņŖĘĒĢśĻ▓ī ĻĖēņä▒ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņŚÉņä£ ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖĻ│╝ ļØ╝ļŗłļ╣äņŻ╝ļ¦ÖņØś ĒÜ©Ļ│╝ļź╝ ļ╣äĻĄÉ ļČäņäØĒĢ£ ņŚ░ĻĄ¼ņŚÉņä£ļÅä ļØ╝ļŗłļ╣äņŻ╝ļ¦ÖņŻ╝ņé¼ĻĄ░ņØ┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņé¼ĻĄ░ļ│┤ļŗż ļ¦Øļ¦ēĒĢśņĢĪņØś ņåīņŗżļÉśĻĖ░Ļ╣īņ¦ĆņØś ĻĖ░Ļ░äņØ┤ ļŹö ņ¦¦Ļ▓ī Ļ▒Ėļ”¼ĻĖ░ļŖö ĒĢśņśĆņ£╝ļéś ĒåĄĻ│äņĀüņ£╝ļĪ£ ņ£ĀņØśĒĢśņ¦ĆļŖö ņĢŖņĢśļŗżļŖö Ļ▓░Ļ│╝Ļ░Ć ņ׳ļŗż[24].
ļŗ╣ļć©ļ¦Øļ¦ēļ│æņ”ØņØ┤ļéś ļ¦Øļ¦ēņżæņŗ¼ņĀĢļ¦źĒÅÉņćäņŚÉņä£ļŖö ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ VEGF ļåŹļÅäĻ░Ć ļ¬ģĒÖĢĒĢśĻ▓ī ļåÆļŗż[32,33]. ĒĢśņ¦Ćļ¦ī ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Ø ĒÖśņ×ÉņŚÉņä£ļŖö ļ░®ņłś ļé┤ VEGF ļåŹļÅäĻ░Ć ņĀĢņāü ļīĆņĪ░ĻĄ░Ļ│╝ ņ░©ņØ┤ļź╝ ļ│┤ņØ┤ņ¦Ć ņĢŖņĢśļŗż[34,35]. ļĢīļ¼ĖņŚÉ ļåÆņØĆ Ļ▓░ĒĢ®ļĀźņØä Ļ░Ćņ¦ä ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖņØś ņØ┤ņĀÉņØä ņāüņćäņŗ£ņ╝░ņØä Ļ▓āņ£╝ļĪ£ ņé¼ļŻīļÉ£ļŗż. Jung et al [26] ļśÉĒĢ£ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Ø ĒÖśņ×ÉņØś ņĢ×ļ░®ņłś ļåŹļÅäĻ░Ć Ļ▒┤Ļ░ĢĒĢ£ ņé¼ļ×īĻ│╝ ļ╣äĻĄÉĒĢśņśĆņØä ļĢī ņ░©ņØ┤Ļ░Ć ņŚåņŚłĻĖ░ ļĢīļ¼ĖņŚÉ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØś ņ╣śļŻīļĪ£ anti-VEGF ņĢĮņĀ£ļź╝ ņé¼ņÜ®ĒĢĀ ļĢī ņĢĮņĀ£ņØś Ļ▓░ĒĢ®ļĀźņØ┤ ņāØļ¼╝ĒĢÖņĀü ĒÖ£ņä▒ļÅäļź╝ Ļ▓░ņĀĢĒĢśļŖö ņŻ╝ņÜö ņÜöņØĖņØ┤ ņĢäļŗłļØ╝Ļ│Ā ļ│┤Ļ│ĀĒĢśņśĆļŗż. ņ╣śļŻīņĢĮņĀ£ņØś ņāØļ¼╝ĒĢÖņĀü ĒÖ£ņä▒ļÅäļŖö ņĢĮņĀ£ņØś Ļ▓░ĒĢ®ļĀź ļ┐Éļ¦ī ņĢäļŗłļØ╝ ļ░śĻ░ÉĻĖ░ņØś ņśüĒ¢źņØä ļ░øļŖöļŗż[36]. ņé¼ļ×īņØś ņĢłĻĄ¼ ļé┤ņŚÉņä£ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖņØś ļ░śĻ░ÉĻĖ░ņŚÉ ļīĆĒĢ£ ņŚ░ĻĄ¼ļŖö ņĢäņ¦ü ņŚåņ¦Ćļ¦ī, ņĢłĻĄ¼ļé┤ ļ░śĻ░ÉĻĖ░ļŖö ļČäņ×Éļ¤ēņŚÉ ņØśĒĢ┤ Ļ▓░ņĀĢļÉśļ»ĆļĪ£ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö(ļČäņ×Éļ¤ē: 149 kDa)ņŚÉ ļ╣äĒĢ┤ ļČäņ×Éļ¤ēņØ┤ ļŹö ņ×æņØĆ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ(ļČäņ×Éļ¤ē: 115 kDa)ņØś ļ░śĻ░ÉĻĖ░Ļ░Ć ļŹö ņ¦¦ņØä Ļ▓āņ£╝ļĪ£ ņāØĻ░üļÉ£ļŗż[37]. ļĢīļ¼ĖņŚÉ ļ░śĻ░ÉĻĖ░Ļ░Ć ļŹö ĻĖĖļŗżļŖö ņØ┤ņĀÉņØä ņ¦Ćļŗī ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖņØ┤ ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖņÖĆ ļ╣äņŖĘĒĢ£ ĒÜ©Ļ│╝ļź╝ ļ│┤ņØĖ Ļ▓āņ£╝ļĪ£ ņé¼ļŻīļÉ£ļŗż.
ļ│Ė ņŚ░ĻĄ¼ļŖö ļ¬ć Ļ░Ćņ¦Ć ĒĢ£Ļ│äņĀÉņØä ņ¦Ćļŗīļŗż. ņÜ░ņäĀ, ĻĖēņä▒Ļ│╝ ļ¦īņä▒ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Ø Ļ░üĻ░üņØś ĻĄ░ņŚÉ ļīĆĒĢśņŚ¼ ņ╣śļŻī ņĢĮņĀ£ Ļ░äņØś ĒÜ©Ļ│╝ļź╝ ļ╣äĻĄÉ ļČäņäØĒĢśņ¦Ć ļ¬╗ĒĢśņśĆļŗż. Hwang et al [38]ņØĆ ļ¦īņä▒ņØś ĒŖ╣ņä▒ņØä Ļ░Ćņ¦ĆļŖö ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØĻĄ░ņØ┤ ĻĖēņä▒ņØś ĒŖ╣ņä▒ņØä Ļ░Ćņ¦ĆļŖö ĻĄ░ļ│┤ļŗż CNVĻ░Ć ļ¦ÄĻ│Ā ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņ×ģņłĀņŚÉ ļīĆĒĢ£ ĒÜ©Ļ│╝Ļ░Ć ļŹö ņóŗņĢśļŗżĻ│Ā ļ│┤Ļ│ĀĒĢśņśĆņ£╝ļ®░, ņØ┤ņ▓śļ¤╝ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØś ĒŖ╣ņä▒ņŚÉ ļö░ļØ╝ ņĢĮņĀ£ņŚÉ ļīĆĒĢ£ ĒÜ©Ļ│╝Ļ░Ć ļŗżļź┤Ļ▓ī ļéśĒāĆļéĀ ņłś ņ׳ļŗż. ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ 6Ļ░£ņøö ņØ┤ņāüņØś Ļ▓ĮĻ│╝ļź╝ ļ│┤ņØ┤ļŖö ļ¦īņä▒ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØ┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦ÖĻĄ░ņŚÉņä£ļŖö 5ļ¬ģ, ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖĻĄ░ņŚÉņä£ļŖö 2ļ¬ģņØ┤ ņ׳ņŚłņ£╝ļéś, ĒÖśņ×ÉņłśĻ░Ć ņĀüņ¢┤ Ļ░ü ĻĄ░ņŚÉ ļö░ļźĖ ņĢĮņĀ£ ĒÜ©Ļ│╝ņØś ļČäņäØņØä ņŗ£Ē¢ēĒĢśņ¦Ć ļ¬╗ĒĢśņśĆĻ│Ā, optical coherence tomographyļ¦īņ£╝ļĪ£ Ļ▓ĮĻ│╝ Ļ┤Ćņ░░ņØä ĒĢśņśĆĻĖ░ņŚÉ ņł©ņ¢┤ ņ׳ņØä Ļ░ĆļŖźņä▒ņØ┤ ņ׳ļŖö CNVļź╝ ņÖäļ▓ĮĒĢśĻ▓ī ļ░░ņĀ£ĒĢśņ¦Ć ļ¬╗ĒĢśņśĆņØä Ļ░ĆļŖźņä▒ņØ┤ ņ׳ļŗż. ļæśņ¦ĖļĪ£ ļ╣äĻĄÉņĀü ļŗ©ĻĖ░Ļ░äņØś ņŚ░ĻĄ¼ļĪ£ ņ╣śļŻī ĒÜ©Ļ│╝Ļ░Ć ņóŗņĢä ļ│┤ņØ╝ ņłś ņ׳ļŖö ļ╣äļÜżļ”╝Ļ│╝ ņĢĮņĀ£ļź╝ ņØ┤ņżæļ¦╣Ļ▓Ćļ░®ņŗØņØ┤ ņĢäļŗī ĒÖśņ×ÉĻ░Ć ņ¦üņĀæ ņäĀĒāØĒĢ©ņ£╝ļĪ£ņŹ© Ēæ£ļ│ĖņäĀņĀĢĒÄĖĒīīņØś ļ╣äļÜżļ”╝ņØ┤ ļ░£ņāØĒĢśņśĆņØä Ļ░ĆļŖźņä▒ņØ┤ ņ׳ļŗż. ņģŗņ¦ĖļĪ£ ĒøäĒ¢źņĀüņØĖ ņŚ░ĻĄ¼ņØ┤ļ®░ ļīĆņĪ░ĻĄ░ņØ┤ ņŚåļŗżļŖö ĒĢ£Ļ│äĻ░Ć ņ׳ļŗż. ĻĘĖļ¤¼ļéś ļ│Ė ņŚ░ĻĄ¼ļŖö ņ▓śņØī ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”Øņ£╝ļĪ£ ņ¦äļŗ©ļ░øņØĆ ĒÖśņ×ÉņŚÉņä£ ņ▓½ ņ╣śļŻīņŚÉ ļīĆĒĢ£ ļ╣äĻĄÉ ļČäņäØņØ┤ļ®░, anti-VEGF agent ņóģļźśņŚÉ ļö░ļźĖ ņ╣śļŻī ĒÜ©Ļ│╝ņØś ņĀĢļÅäļź╝ ļ╣äĻĄÉĒĢśņśĆļŗżļŖö ņĀÉņŚÉ ņØśņØśļź╝ ļæöļŗż. Ļ▓░Ļ│╝ņĀüņ£╝ļĪ£ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØĒÖśņ×ÉņŚÉņä£ ņ£Āļ”¼ņ▓┤Ļ░Ģļé┤ ļ▓Āļ░öņŗ£ņŻ╝ļ¦Ö ņŻ╝ņ×ģņłĀ ļśÉļŖö ņĢĀĒöīļ”¼ļ▓äņģēĒŖĖ ņŻ╝ņ×ģņłĀ ļ¬©ļæÉ ņżæņŗ¼ņןņĢĪļ¦źļØĮļ¦Øļ¦ēļ│æņ”ØņØś ĻĄ¼ņĪ░ņĀü ļ░Å ĻĖ░ļŖźņĀü ĒÜīļ│ĄņŚÉ ĒÜ©Ļ│╝Ļ░Ć ņ׳ņŚłņØīņØä ĒÖĢņØĖĒĢĀ ņłś ņ׳ņŚłņ£╝ļ®░, ĻĖ░ļŖźņĀü ĻĘĖļ”¼Ļ│Ā ĒĢ┤ļČĆĒĢÖņĀü ĒśĖņĀäņØś ņĀĢļÅä, ņ╣śļŻīĻ╣īņ¦Ć ņŗ£Ē¢ēļÉ£ ņŻ╝ņé¼ Ēܤņłś ļ░Å ĻĖ░Ļ░äņŚÉ ņĢĮņĀ£ņØś ņóģļźśļŖö ņśüĒ¢źņØä ļ»Ėņ╣śņ¦Ć ņĢŖņĢśļŗż. ņČöĒøä ĒĢ£Ļ│äņĀÉļōżņØä ļ│┤ņÖäĒĢ£ ļīĆĻĘ£ļ¬©ņØś ņןĻĖ░Ļ░ä ņČöņĀü Ļ┤Ćņ░░ ņŚ░ĻĄ¼Ļ░Ć ņØ┤ļŻ©ņ¢┤ņ¦Ćļ®┤ ņ╣śļŻī ļ░®ņ╣©ņØä ņäĖņÜ░ļŖö ļŹ░ņŚÉ ļŹö ļÅäņøĆņØ┤ ļÉĀ Ļ▓āņ£╝ļĪ£ ņŚ¼Ļ▓©ņ¦äļŗż.
Figure┬Ā1.
Comparison of best-corrected visual acuity (BCVA) and subfoveal choroidal thickness (SCT) according to the group of injection type before and 3 months after treatment. Gray bar and gray line mean bevacizumab group, Black bar and black line mean aflibercept group. *Statistically significance is p < 0.05 with baseline.
Table┬Ā1.
Baseline characteristics according to the group of injection type
Values are presented as mean ┬▒ standard deviation unless otherwise indicated.
M/F = male/female; BCVA = best-corrected visual acuity; logMAR = logarithm of the minimal angle of resolution; SCT = subfoveal choroidal thickness.
p-value for two-group comparison by *independent t-test; ŌĆĀchi-square test, and ŌĆĪMann-Whitney U test.
REFERENCES
1) Chhablani J, Kozak I, Pichi F, et al. Outcomes of treatment of choroidal neovascularization associated with central serous chorioretinopathy with intravitreal antiangiogenic agents. Retina 2015;35:2489-97.


2) Lim SJ, Roh MI, Kwon OW. Intravitreal bevacizumab injection for central serous chorioretinopathy. Retina 2010;30:100-6.


3) Bae SH, Heo JW, Kim C, et al. A randomized pilot study of low-fluence photodynamic therapy versus intravitreal ranibizumab for chronic central serous chorioretinopathy. Am J Ophthalmol 2011;152:784-92.e2.


4) Chung YR, Kim JW, Song JH, et al. Twelve-month efficacy of intravitreal bevacizumab injection for chronic, atypical, or recurrent central serous chorioretinopathy. Retina 2019;39:134-42.


5) Schaal KB, Hoeh AE, Scheuerle A, et al. Intravitreal bevacizumab for treatment of chronic central serous chorioretinopathy. Eur J Ophthalmol 2009;19:613-7.


6) Artunay O, Yuzbasioglu E, Rasier R, et al. Intravitreal bevacizumab in treatment of idiopathic persistent central serous chorioretinopathy: a prospective, controlled clinical study. Curr Eye Res 2010;35:91-8.


7) Inoue M, Kadonosono K, Watanabe Y, et al. Results of one-year follow-up examinations after intravitreal bevacizumab administration for chronic central serous chorioretinopathy. Ophthalmologica 2011;225:37-40.


8) Broadhead GK, Chang A. Intravitreal aflibercept for choroidal neovascularisation complicating chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol 2015;253:979-81.


9) Pitcher JD 3rd, Witkin AJ, DeCroos FC, Ho AC. A prospective pilot study of intravitreal aflibercept for the treatment of chronic central serous chorioretinopathy: the CONTAIN study. Br J Ophthalmol 2015;99:848-52.


10) Aydin E. The efficacy of intravitreal bevacizumab for acute central serous chorioretinopathy. J Ocul Pharmacol Ther 2013;29:10-3.


11) Seong HK, Bae JH, Kim ES, et al. Intravitreal bevacizumab to treat acute central serous chorioretinopathy: short-term effect. Ophthalmologica 2009;223:343-7.


12) Torres-Soriano ME, Garc├Ła-Aguirre G, Kon-Jara V, et al. A pilot study of intravitreal bevacizumab for the treatment of central serous chorioretinopathy (case reports). Graefes Arch Clin Exp Ophthalmol 2008;246:1235-9.


13) Kweon EY. Factors influencing the effect of the intravitreal bevacizumab injection in patients with central serous chorioretinopathy. J Korean Ophthalmol Soc 2014;55:391-5.

14) Carmeliet P, Moons L, Luttun A, et al. Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med 2001;7:575-83.


15) Stewart MW. Aflibercept (VEGF Trap-eye): the newest anti-VEGF drug. Br J Ophthalmol 2012;96:1157-8.


16) Binder S. Loss of reactivity in intravitreal anti-VEGF therapy: tachyphylaxis or tolerance? Br J Ophthalmol 2012;96:1-2.


17) Gasperini JL, Fawzi AA, Khondkaryan A, et al. Bevacizumab and ranibizumab tachyphylaxis in the treatment of choroidal neovascularisation. Br J Ophthalmol 2012;96:14-20.


18) Radke N, Kalamkar C, Mukherjee A, Radke S. Intravitreal ziv-aflibercept in treatment of naïve chronic central serous chorioretinopathy related choroidal neovascular membrane. Case Rep Ophthalmol Med 2017;2017:5036248.



19) Nicholson B, Noble J, Forooghian F, Meyerle C. Central serous chorioretinopathy: update on pathophysiology and treatment. Surv Ophthalmol 2013;58:103-26.



20) Yannuzzi LA. Central serous chorioretinopathy: a personal perspective. Am J Ophthalmol 2010;149:361-3.


21) Manayath GJ, Ranjan R, Karandikar SS, et al. Central serous chorioretinopathy: current update on management. Oman J Ophthalmol 2018;11:200-6.



22) Kim M, Lee SC, Lee SJ. Intravitreal ranibizumab for acute central serous chorioretinopathy. Ophthalmologica 2013;229:152-7.


23) Park SU, Lee SJ, Kim M. Intravitreal anti-vascular endothelial growth factor versus observation in acute central serous chorioretinopathy: one-year results. Korean J Ophthalmol 2014;28:306-13.



24) Tekin K, Sekeroglu MA, Cankaya AB, et al. Intravitreal bevacizumab and ranibizumab in the treatment of acute central serous chorioretihopathy: a single center retrospective study. Semin Ophthalmol 2018;33:265-70.


25) Koh KM, Kim JY, Kim JW, Choi MJ. The efficacy of intravitreal bevacizumab injection in patients with acute central serous chorioretinopathy. J Korean Ophthalmol Soc 2012;53:781-5.

26) Jung BJ, Lee K, Park JH, Lee JH. Chorioretinal response to intravitreal aflibercept injection in acute central serous chorioretinopathy. Int J Ophthalmol 2019;12:1865-71.



27) Alishiri A, Jadidi K, Mosavi SA, Torabi H. Intravitreal bevacizumab administration for the treatment of chronic central serous chorioretinopathy. J Curr Ophthalmol 2019;31:406-10.



28) Tekin K, Cakar Ozdal P, Gulpamuk B, Yasin Teke M. Intravitreal aflibercept therapy in eyes with chronic central serous chorioretihopathy: short term results. Arch Soc Esp Oftalmol (Engl Ed) 2018;93:315-23.


29) Wells JA, Glassman AR, Ayala AR, et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema: two-year results from a comparative effectiveness randomized clinical trial. Ophthalmology 2016;123:1351-9.

30) Heier JS, Bressler NM, Avery RL, et al. Comparison of aflibercept, bevacizumab, and ranibizumab for treatment of diabetic macular edema: extrapolation of data to clinical practice. JAMA Ophthalmol 2016;134:95-9.


31) Lotfy A, Solaiman KAM, Abdelrahman A, Samir A. Efficacy and frequency of intravitreal aflibercept versus bevacizumab for macular edema secondary to central retinal vein occlusion. Retina 2018;38:1795-800.


32) Noma H, Funatsu H, Mimura T, et al. Vitreous levels of interleukin- 6 and vascular endothelial growth factor in macular edema with central retinal vein occlusion. Ophthalmology 2009;116:87-93.


33) Funatsu H, Yamashita H, Sakata K, et al. Vitreous levels of vascular endothelial growth factor and intercellular adhesion molecule 1 are related to diabetic macular edema. Ophthalmology 2005;112:806-16.


34) Lim JW, Kim MU, Shin MC. Aqueous humor and plasma levels of vascular endothelial growth factor and interleukin-8 in patients with central serous chorioretinopathy. Retina 2010;30:1465-71.


35) Shin MC, Lim JW. Concentration of cytokines in the aqueous humor of patients with central serous chorioretinopathy. Retina 2011;31:1937-43.


36) Bodla MA, Bodla AA, Kazmi SM, Moazzam A. Effects of intravitreal aflibercept in patients with central serous chorioretinopathy. Pak J Ophthalmol 2020;36:402-7.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print