|
 |
| J Korean Ophthalmol Soc > Volume 62(4); 2021 > Article |
|
ĻĄŁļ¼Ėņ┤łļĪØ
ļ¬®ņĀü
0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ ņĢłņĢĮņØś ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅäņŚÉ ļ»Ėņ╣śļŖö ņśüĒ¢źļĀź ņØĖņ×Éļź╝ ĒāÉņāēĒĢśĻ│Āņ×É ĒĢśņśĆļŗż.
ļīĆņāüĻ│╝ ļ░®ļ▓Ģ
0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝(Ikervis┬«)ņØä ņ▓śļ░®ļ░øņØĆ ĒÖśņ×É ņżæ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅäļź╝ ĒÅēĻ░ĆĒĢśļŖö EDSQ ņäżļ¼Ėņ¦Ć(ņĀÉņĢłĻ░É ĒÅēĻ░Ć: ņ¦łļ¼Ė 1-4, ņł£ņØæļÅä ĒÅēĻ░Ć: ņ¦łļ¼Ė 5-7)ļź╝ ņ×æņä▒ĒĢ£ ĒÖśņ×Éļź╝ ļīĆņāüņ£╝ļĪ£ ĒĢśņśĆļŗż. ņØ┤ļōż ņżæ ņ┤łĻĖ░ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅäņŚÉ ņśüĒ¢źņØä ļ»Ėņ╣śļŖö ņØĖņ×Éļź╝ ļČäņäØĒĢśĻĖ░ ņ£äĒĢ┤ ņĀÉņĢł ņ┤łĻĖ░ 3Ļ░£ņøö ļé┤ ņĄ£ņ┤ł ņäżļ¼ĖņØä ņŗ£Ē¢ēĒĢ£ ĒÖśņ×ÉĻĄ░(ņĮöĒśĖĒŖĖ 1)ņŚÉņä£ Ļ░ü ņ¦łļ¼ĖņØś ņĀÉņłśņÖĆ ņŚ░ļĀ╣, ņä▒ļ│ä, ņĀÉņĢł ĻĖ░Ļ░ä, ļ¦łņØ┤ļ┤äņāś ĻĖ░ļŖźņןņĢĀ, ļłłļ¼╝ ļČäļ╣äļ¤ē, Ļ░üļ¦ēņ¦ĆĻ░ü ņŚŁņ╣ś, Ļ░üļ¦ēļ»Ėļ×Ć, ņĢłĻĄ¼Ēæ£ļ®┤ņŚ╝ņāēņ¦Ćņłś, ļłłļ¼╝ ļé┤ matrix metalloproteinase 9 (MMP-9) ļ░£Ēśä ļō▒ĻĖē ļ░Å ņĢłĻĄ¼ Ēæ£ļ®┤ņ¦łĒÖśņ¦Ćņłś Ļ░ä ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļČäņäØĒĢśņśĆļŗż. ļśÉĒĢ£, ņĀÉņĢł ņ¦ĆņåŹņŚÉ ļö░ļźĖ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅäņØś ļ│ĆĒÖöļź╝ ĒÅēĻ░ĆĒĢśĻĖ░ ņ£äĒĢ┤ 2ĒÜī ņØ┤ņāüņØś ņäżļ¼ĖņØä ņŗ£Ē¢ēĒĢ£ ĒÖśņ×ÉĻĄ░(ņĮöĒśĖĒŖĖ 2)ņŚÉņä£ Ļ░ü ņ¦łļ¼Ė ņĀÉņłśļōżņØś ņŗ£Ļ░äņŚÉ ļö░ļźĖ ļ│ĆĒÖöļź╝ ļČäņäØĒĢśņśĆļŗż.
Ļ▓░Ļ│╝
ņĮöĒśĖĒŖĖ 1 (39ļ¬ģ, 74ņĢł)ņŚÉņä£ ņ¦łļ¼Ė 1-4ņØś ņĀÉņĢł ņŗ£ ļČłĒÄĖĻ░É ņĀÉņłśļōżņØĆ ņŚ¼ņä▒ņŚÉņä£ ņ£ĀņØśĒĢśĻ▓ī ļŹö ļåÆņĢśĻ│Ā, ņĢłņĢĮ ņĀÉņĢł ĻĖ░Ļ░ä ļ░Å ņŚ░ļĀ╣Ļ│╝ļŖö ņØīņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝, ļłłļ¼╝ ļČäļ╣äļ¤ē, Ļ░üļ¦ēļ»Ėļ×Ć, ņĢłĻĄ¼Ēæ£ļ®┤ņŚ╝ņāēņ¦Ćņłś ļ░Å ļłłļ¼╝ ļé┤ MMP-9 ļ░£Ēśä ļō▒ĻĖēĻ│╝ļŖö ņ¢æņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņśĆļŗż. ļłłļ¼╝ ļé┤ MMP-9 ļ░£Ēśä ļō▒ĻĖēņØ┤ ļåÆņØäņłśļĪØ ņ¦łļ¼Ė 5ņØś ņĢĮņĀ£ ņł£ņØæļÅä ņĀÉņłśĻ░Ć ņ£ĀņØśĒĢśĻ▓ī ļé«ņĢśļŗż. ņĮöĒśĖĒŖĖ 2 (34ļ¬ģ)ņŚÉņä£ ņĀÉņĢł ņŗ£ ļČłĒÄĖĻ░É ņĀÉņłśļōżņØĆ ļłäņĀü ņĀÉņĢł ĻĖ░Ļ░äņØ┤ ĻĖĖņ¢┤ņ¦Ćļ®┤ņä£ Ļ░ÉņåīĒĢśļŖö Ļ▓ĮĒ¢źņØä ļ│┤ņśĆļŗż.
ABSTRACT
Purpose
We investigated the contributing factors affecting the ocular discomfort on instillation and compliance of 0.1% cyclosporin A (CsA) cationic nanoemulsion eye drops.
Methods
We enrolled patients who were prescribed 0.1% CsA eye drops (Ikervis┬«) and who filled out an eye drop satisfaction questionnaire to assess ocular discomfort on instillation (questions Q1-Q4) and compliance of eye drops (Q5-Q7). First, to identify the contributing factors affecting the early ocular discomfort of 0.1% CsA instillation and compliance, Q1-Q7 scores were correlated with respect to age, sex, instillation period duration, meibomian gland dysfunction, tear secretion, corneal sensitivity, corneal erosions, Sj├Čgren's International Collaborative Clinical Alliance ocular staining score (OSS), tear matrix metalloproteinase 9 (MMP-9), and ocular surface disease index in patients who completed the first questionnaire within the first 3 months of instillation of 0.1% CsA (referred to as Cohort 1). Second, to evaluate the change in ocular discomfort on instillation and compliance, along with the prolonged instillation of 0.1% CsA, the changes in individual scores for questions Q1-Q7 were analyzed in patients who completed at least two or more serial questionnaires (corresponding to Cohort 2).
Results
In Cohort 1 (74 eyes in 39 patients), the scores for ocular discomfort on instillation (Q1-Q4) were higher in females and correlated negatively with the instillation period duration and age and positively with tear secretion, corneal erosions, OSS, and tear MMP-9 grades. The higher the grade of tear MMP-9, the lower the compliance score of Q5. In Cohort 2 (34 patients), the scores for ocular discomfort on instillation tended to decrease as the cumulative instillation period lengthened.
ņĄ£ĻĘ╝ ņĢīļĀżņ¦ä Tear Film & Ocular Surface Society (TFOS) Dry Eye WorkShop (DEWS) II ļ”¼ĒżĒŖĖņŚÉ ļö░ļź┤ļ®┤, ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”ØņØĆ ļłłļ¼╝ņĖĄņØś ļČłņĢłņĀĢņä▒, ļłłļ¼╝ ļé┤ Ļ│Āņé╝Ēł¼ņĢĢ, ņĢłĻĄ¼ Ēæ£ļ®┤ņØś ņŚ╝ņ”Ø ļ░Å ņåÉņāü, Ļ░ÉĻ░üņŗĀĻ▓Į ņØ┤ņāüņØś ļŗżņØĖņä▒ ļ│æĒā£ņāØļ”¼Ļ░Ć Ļ┤ĆņŚ¼ĒĢśļŖö ņ¦łĒÖśĻĄ░ņØ┤ļŗż[1]. ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”ØņØĆ ņĢłĻĄ¼ ņ×ÉĻĘ╣Ļ░É, ĒåĄņ”Ø, Ļ▒┤ņĪ░Ļ░É, ņ×æņŚ┤Ļ░É, ņØ┤ļ¼╝Ļ░É, ņŗ£ļĀź ļ│ĆļÅÖ ļō▒ņØś ļŗżņ¢æĒĢ£ ņ”ØņāüņØä ņ£Āļ░£ĒĢśļ®░[2], ņżæņ”ØņØ┤ļéś ļ¦īņä▒ ĒÖśņ×ÉņŚÉņä£ļŖö Ļ▓░ļ¦ēļ░śĒØö, ņŗżļ¬©ņ¢æ Ļ░üļ¦ēņŚ╝, Ļ░üļ¦ēņāüĒö╝ Ļ╣īņ¦É Ēś╣ņØĆ ĻČżņ¢æņØä ņ┤łļלĒĢśĻĖ░ ļĢīļ¼ĖņŚÉ ņéČņØś ņ¦łņØä ļ¢©ņ¢┤ļ£©ļ”¼Ļ│Ā ņØśļŻīļ╣ä ļČĆļŗ┤ņØä Ļ░Ćņżæņŗ£ĒéżĻ▓ī ļÉ£ļŗż[3].
ņŚ░ņćäņĀüņØĖ ņŚ╝ņ”Ø ļ░śņØæņØ┤ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ļ│æņØĖņØś ņĢģņł£ĒÖś Ļ│Āļ”¼ņŚÉ ņ׳ņ¢┤ ĒĢĄņŗ¼ņĀüņØĖ ņŚŁĒĢĀņØä ņ░©ņ¦ĆĒĢśĻ│Ā ņ׳ĻĖ░ ļĢīļ¼ĖņŚÉ[4] ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”ØņØś ņ╣śļŻīļĪ£ ņŖżĒģīļĪ£ņØ┤ļō£ ļśÉļŖö ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░(Cyclosporine A) ļō▒ņØś ĒĢŁņŚ╝ņ”Øņä▒ ņĀ£ņĀ£ņØś ņĀÉņĢłņØ┤ ļäÉļ”¼ ņØ┤ņÜ®ļÉśņ¢┤ ņÖöļŗż. ĻĘĖņżæ ņĀÉņĢł ņĮöļź┤Ēŗ░ņĮöņŖżĒģīļĪ£ņØ┤ļō£ļŖö ņ”ØņāüĻ│╝ ņĢłĻĄ¼ Ēæ£ļ®┤ņØś ļ╣äņĀĢņāü ņåīĻ▓¼ņØä ĒśĖņĀäņŗ£Ēé¼ ņłś ņ׳ņ¦Ćļ¦ī, Ļ│ĀņĢłņĢĢ, ļ░▒ļé┤ņן ļ░£ņāØ ļō▒ņØś ļČĆņ×æņÜ® ļĢīļ¼ĖņŚÉ ĒśäņŗżņĀüņ£╝ļĪ£ ņןĻĖ░Ļ░ä ņé¼ņÜ®ĒĢśĻĖ░ ņ¢┤ļĀĄļŗż[5]. ņØ┤ņŚÉ ņŖżĒģīļĪ£ņØ┤ļō£ ņĀ£ņĀ£ņØś ļīĆņĢłņ£╝ļĪ£ņä£ 2003ļģä 0.05% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņØīņØ┤ņś© ņŚÉļ®ĆņĀ╝(Restasis┬«; Allergan, Irvine, CA, USA)ņØ┤ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ņ╣śļŻīņĀ£ļĪ£ņä£ ļ»ĖĻĄŁ ņŗØĒÆłņØśņĢĮĻĄŁņØś ņŖ╣ņØĖņØä ļ░øņĢśĻ│Ā, ņØ┤Ēøä ņןĻĖ░Ļ░ä ņĢłņĀäņä▒ņØä Ļ░Ćņ¦ä ņĢĮļ¼╝ļĪ£ņä£ ļäÉļ”¼ ņØ┤ņÜ®ļÉśņ¢┤ ņÖöļŗż[5,6]. ņĄ£ĻĘ╝ 2015ļģäņŚÉļŖö ņ¢æņØ┤ņś© ņŚÉļ®ĆņĀ╝ ĒśĢĒā£ņØś 0.1% ņé¼ņØ┤Ēü┤ļĪ£ļ”░ ņĀÉņĢłņĀ£(Ikervis┬«, Santen SAS, Evry, France)Ļ░Ć ņ£Āļ¤ĮņŚÉņä£ ņĄ£ņ┤ł ņČ£ņŗ£ļÉśņŚłĻ│Ā ņżæļō▒ļÅä ļśÉļŖö ņżæņ”ØņØś ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÖśņ×ÉņŚÉņä£ ĒÜ©Ļ│╝ņĀüņØ┤ļØ╝Ļ│Ā ņĢīļĀżņĪīļŗż[7-9]. 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ņØĆ ĻĖ░ņĪ┤ņØś 0.05% ņØīņØ┤ņś© ņĀ£ņĀ£ņÖĆ ļŗ¼ļ”¼ ņ¢æņØ┤ņś©ņØä ļØäĻĖ░ ļĢīļ¼ĖņŚÉ ņØīņØ┤ņś© ņäĖĒżļ¦ēņŚÉ ļŹö ņēĮĻ▓ī ĻĘ╝ņĀæĒĢĀ ņłś ņ׳Ļ│Ā[10], ļåŹļÅä ņŚŁņŗ£ ĻĖ░ņĪ┤ ļīĆļ╣ä 2ļ░░ļĪ£ ļåÆņśĆĻĖ░ ļĢīļ¼ĖņŚÉ ņĢłĻĄ¼ Ēæ£ļ®┤ņŚÉņä£ ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ņØś ņ×öņĪ┤ ņŗ£Ļ░ä ļ░Å ņāØņ▓┤ ņØ┤ņÜ®ļźĀņØä Ē¢źņāüņŗ£ĒéżĻ│Ā ņĀÉņĢł Ēܤņłś ņŚŁņŗ£ 1ņØ╝ 1ĒÜīļĪ£ ņżäņØ╝ ņłś ņ׳Ļ▓ī ļÉśņŚłļŗż[11-14].
ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ņØĆ ļČäņ×Éļ¤ēņØ┤ Ēü¼Ļ│Ā ņ╣£ņ¦Ćņ¦łņä▒(lipophilic) ļ¼╝ņ¦łļĪ£ņä£ ļ¼╝ņŚÉ ņל ļģ╣ņ¦Ć ņĢŖļŖöļŗż[15]. ņØ┤ ļĢīļ¼ĖņŚÉ ņāüņÜ® ņĢłņĢĮņ£╝ļĪ£ ņāØņé░ ņŗ£ Ēö╝ļ¦łņ×Éņ£Ā(castor oil) Ēś╣ņØĆ ņżæņćäĒŖĖļ”¼ĻĖĆļ”¼ņäĖļ”¼ļō£(mediumchain tryclyceride) ļō▒ņØś ņ£ĀĻĖ░ ņÜ®ļ¦żņŚÉ ļģ╣ņØĖ Ēøä Ļ│äļ®┤ĒÖ£ņä▒ņĀ£(surfactant)ņØś ļÅäņøĆņØä ļ░øņĢä ļ¼╝ ņåŹņŚÉņä£ ņŚÉļ®ĆņĀ╝ Ēś╝ĒĢ®ļÉ£ ĒśĢĒā£(oil-in-water emulsion)ļĪ£ ņāØņé░ĒĢśĻ▓ī ļÉśļŖöļŹ░[12], ņØ┤ļ¤¼ĒĢ£ ļÅģĒŖ╣ĒĢ£ ĒśĢĒā£ņØś ņä▒ļČäņ£╝ļĪ£ ņØĖĒĢśņŚ¼ ņĀÉņĢł ņŗ£ ĒåĄņ”Ø, ĒÖöļüłĻ▒░ļ”╝, ņŗ£ņĢ╝ ĒØÉļ”╝ ļō▒ņØś ļČłĒÄĖĻ░ÉņØä ņ£Āļ░£ĒĢ£ļŗż[10,16]. ņĄ£ĻĘ╝ ņČ£ņŗ£ļÉ£ 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ ņĢłņĢģņĀ£ ņŚŁņŗ£ ņŚ¼ļ¤¼ ņ×äņāü ņŗ£ĒŚśņŚÉņä£ ņĀÉņĢł ņŗ£ ĒåĄņ”Ø Ēś╣ņØĆ ņ×ÉĻĘ╣Ļ░ÉņØ┤ 13.0-29.2%ņØś ĒÖśņ×ÉņŚÉņä£ ļ░£ņāØĒĢ£ļŗżĻ│Ā ļ│┤Ļ│ĀļÉśņŚłļŗż[8,9,17].
ņĢłņĢĮ ņĀÉņĢł ņŗ£ņØś ļČłĒÄĖĻ░ÉņØĆ ņØ╝ļ░śņĀüņ£╝ļĪ£ ņĢĮņĀ£ ņł£ņØæļÅäļź╝ ļ¢©ņ¢┤ļ£©ļ”¼Ļ│Ā Ļ▓░Ļ│╝ņĀüņ£╝ļĪ£ ņ╣śļŻī ĒÜ©Ļ│╝ ņŚŁņŗ£ Ļ░Éņåīņŗ£Ēé©ļŗż[18]. 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņĀÉņĢłņĀ£ļŖö ņŗżņĀ£ ņżæļō▒ļÅä ņØ┤ņāüņØś ņŗ¼ĒĢ£ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”ØņŚÉņä£ ņŻ╝ļĪ£ ņé¼ņÜ®ĒĢśĻ▓ī ļÉśļ®░, ņØ┤ļōż ĒÖśņ×ÉļōżņØĆ ņĢĮņĀ£ņØś ņĀüņĀłĒĢ£ ņé¼ņÜ®ņØ┤ ņä▒Ļ│ĄņĀüņØĖ ņ╣śļŻīņŚÉ ņ׳ņ¢┤ ņżæņÜöĒĢśļŗż. ņØ┤ņŚÉ ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ļŖö 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ ņé¼ņÜ® ņŗ£ ņé¼ņĀäņŚÉ ĒÖśņ×É ņäżļ¬ģņŚÉ ļÅäņøĆņØ┤ ļÉĀ ņłś ņ׳ļŖö ņĀüņĀłĒĢ£ ņ¦Ćņ╣©ņØä ļ¦łļĀ©ĒĢśĻĖ░ ņ£äĒĢ┤ ņĢĮņĀ£ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅäņÖĆ ņŚ░Ļ┤Ćņä▒ņØ┤ ņ׳ļŖö ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ļ░Å ņĢłĻĄ¼ Ēæ£ļ®┤ ņŚ╝ņ”Ø ņ¦ĆĒæ£ļōżņØä ņĢīņĢäļ│┤Ļ│Āņ×É ĒĢśņśĆļŗż.
ļ│Ė ņŚ░ĻĄ¼ļŖö ĒøäĒ¢źņĀü ņØśļ¼┤ĻĖ░ļĪØ ņĪ░ņé¼ ņŚ░ĻĄ¼ļĪ£ņä£, ļ│ĖņøÉņØś ņŚ░ĻĄ¼ņ£żļ”¼ņŗ¼ņØśņ£äņøÉĒÜī(Institutional Review Board [IRB])ņØś ņŖ╣ņØĖĒĢś(IRB ņŖ╣ņØĖ ļ▓łĒśĖ: 2004-007-19310) ĒŚ¼ņŗ▒ĒéżņäĀņ¢ĖņŚÉ ņ×ģĻ░üĒĢśņŚ¼ ņłśĒ¢ēļÉśņŚłļŗż.
2019ļģä 9ņøöļČĆĒä░ 2020ļģä 8ņøö ņé¼ņØ┤ņŚÉ ļłłļ¼╝ ļé┤ matrix metalloproteinase (MMP)-9 ļ░£ĒśäņØ┤ ņ¢æņä▒ņØĖ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø, ļ¦łņØ┤ļ┤äņāś ĻĖ░ļŖźņןņĢĀ ļ░Å ņŻ╝ļ│ĆļČĆ Ļ░üļ¦ēĻČżņ¢æ, ņć╝ĻĘĖļĀīņ”ØĒøäĻĄ░, ņŗĀĻ▓Įņśüņ¢æņä▒ Ļ░üļ¦ēņŚ╝, ņŖżĒŗ░ļĖÉņŖż-ņĪ┤ņŖ©ņ”ØĒøäĻĄ░, ļłł ĒØēĒä░ņä▒ ņ£Āņ▓£Ēżņ░Į(ocular cicatricial pemphigoid), ļłł ņŻ╝ņé¼(rosacea), ļłł ņØ┤ņŗØĒÄĖļīĆņłÖņŻ╝ ļ░śņØæ ļĢīļ¼ĖņŚÉ 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝(Ikervis┬«, Santen SAS)ņØä ĒĢśļŻ© 1ĒÜī ņÜ®ļ▓Ģņ£╝ļĪ£ ņ▓śļ░® ļ░øņØĆ ĒÖśņ×Éļōż ņżæ ņĄ£ņåī 2ņŻ╝ ņØ┤ņāü ņĀÉņĢłĒĢśņśĆĻ│Ā, ņĄ£ņåī 1ĒÜī ņØ┤ņāü ņĢĮņĀ£ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅä ņäżļ¼Ė ĒÅēĻ░Ćļź╝ ņłśĒ¢ēĒĢ£ ĒÖśņ×ÉļōżņØä ņĀäņ▓┤ ļīĆņāüņ£╝ļĪ£ ĒĢśņśĆļŗż.
ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ ņé¼ņÜ®ĒĢ£ ņäżļ¼Ėņ¦ĆļŖö Ļ│╝Ļ▒░ ļģ╣ļé┤ņן ņĢĮņĀ£ņØś ņĀÉņĢł ņŗ£ ļČłĒÄĖĻ░ÉņØä ĒÅēĻ░ĆĒĢśĻĖ░ ņ£äĒĢ┤ ņśüņ¢┤ņÖĆ ļČłņ¢┤ļĪ£ ļÅÖņŗ£ņŚÉ Ļ░£ļ░£ļÉ£ Eye-drop satisfaction questionnaire (EDSQ) ņäżļ¼Ėņ¦Ć[19,20]ņØś ņäżļ¼Ė ņäĖļČĆ ņŻ╝ņĀ£ļź╝ ĻĘ╝Ļ░äņ£╝ļĪ£ ĒĢ£ĻĖĆļĪ£ ĒÆĆņ¢┤ņä£ ņŹ©ņä£ ļ¦łļĀ©ĒĢśņśĆļŗż. EDSQ ņäżļ¼Ėņ¦ĆļŖö ņĢłĻ│╝ņĀü ņĀÉņĢł ņ╣śļŻī ņżæņØĖ ĒÖśņ×ÉņŚÉņä£ ņ¦łĒÖśņŚÉ ļīĆĒĢ£ ņāØĻ░ü, ņ╣śļŻī ļ¦īņĪ▒ļÅä, ņĀÉņĢłĻ░É ļ░Å ņ╣śļŻī ņł£ņØæļÅäļź╝ ņóģĒĢ®ņĀüņ£╝ļĪ£ ņ×ÉĻ░Ć ĒÅēĻ░ĆĒĢśļŖö ņäżļ¼Ėņ¦ĆļĪ£ņä£, 1) ņ╣śļŻīņŚÉ ļīĆĒĢ£ Ļ▒▒ņĀĢ ĒÅēĻ░Ć ņśüņŚŁ(concerns about treatment), 2) ņ¦łļ│æņŚÉ ļīĆĒĢ£ Ļ▒▒ņĀĢ ļ░Å ņÜ░ļĀż ĒÅēĻ░Ć ņśüņŚŁ(concerns about disease), 3) ĒÖśņ×É-ņØśņé¼ Ļ░ä Ļ┤ĆĻ│ä ļ¦īņĪ▒ļÅä ĒÅēĻ░Ć ņśüņŚŁ(satisfaction with patient-clinician relationship), 4) ņ╣śļŻī Ļ│╝ņĀĢņŚÉņä£ņØś ĻĖŹņĀĢņĀüņØĖ ļ»┐ņØī ĒÅēĻ░Ć ņśüņŚŁ(positive belief), 5) ņ╣śļŻī ĒÄĖļ”¼ņä▒ ĒÅēĻ░Ć ņśüņŚŁ(treatment convenience), 6) ņ×ÉĻ░Ć ņł£ņØæļÅä ĒÅēĻ░Ć ņśüņŚŁ(self-declared compliance)ņØś 6Ļ░£ ņśüņŚŁņŚÉņä£ ņ┤Ø 21Ļ░£ņØś ņäżļ¼ĖņØä ņŗ£Ē¢ēĒĢśņŚ¼ ņĀÉņłśļź╝ ļ¦żĻĖ┤ļŗż[19]. ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ļŖö EDSQ ņäżļ¼Ėņ¦ĆņØś 6Ļ░£ ņśüņŚŁ ņżæ ņĢłņĢĮ ņĀÉņĢłĻ░ÉņØä ĒÅēĻ░ĆĒĢśļŖö ņśüņŚŁņØĖ 1) ņ╣śļŻīņŚÉ ļīĆĒĢ£ Ļ▒▒ņĀĢ ĒÅēĻ░Ć ņśüņŚŁņŚÉņä£ 4Ļ░£ņØś ņ¦łļ¼Ė: ņ¦łļ¼Ė 1) ņĀÉņĢł ņ¦üĒøä ļłłņØ┤ ļČłĒÄĖĒ¢łļŖöņ¦Ć(discomfort), ņ¦łļ¼Ė 2) ņĀÉņĢłĻ░ÉņØ┤ ļČłĒÄĖĒĢĀĻ╣īļ┤É ļ»Ėļ”¼ Ļ▒▒ņĀĢņØ┤ ļÉśņŚłļŖöņ¦Ć(worries about putting things in eyes), ņ¦łļ¼Ė 3) ņĀÉņĢł ņ¦üĒøä ļéśļÅä ļ¬©ļź┤Ļ▓ī ļłłņØ┤ Ļ╣£ļ╣ĪņśĆļŖöņ¦Ć(blinking reflex), ņ¦łļ¼Ė 4) ņĀÉņĢł ļČłĒÄĖĻ░ÉņØ┤ ņŗ¼ņĀü ļČĆļŗ┤ņØ┤ ļÉśņŚłļŖöņ¦Ć(treatment as a burden)ņÖĆ ņĢĮņĀ£ ņé¼ņÜ®ņØś ņł£ņØæļÅäļź╝ ĒÅēĻ░ĆĒĢśļŖö ņśüņŚŁņØĖ 6) ņ×ÉĻ░Ć ņł£ņØæļÅä ĒÅēĻ░Ć ņśüņŚŁņŚÉņä£ 3Ļ░£ņØś ņ¦łļ¼Ė: ņ¦łļ¼Ė 5) ņ¦Ćļé£ 4ņŻ╝Ļ░ä ļéśļ”ä ņל ņĀÉņĢłĒ¢łļŗżĻ│Ā ņāØĻ░üĒĢśļŖöņ¦Ć(self-assessed compliance over the last 4 weeks), ņ¦łļ¼Ė 6) ņĀÉņĢłĒĢśļŖö Ļ▓āņØä ņל ņ×Ŗņ¢┤ļ▓äļĀĖļŖöņ¦Ć(frequency treatment is forgotten in general), ņ¦łļ¼Ė 7) ņØ╝ļČĆļ¤¼ ņĀÉņĢłņØä ņĢłĒĢśĻĖ░ļÅä Ē¢łļŖöņ¦Ć(frequency of voluntary break from treatment)ļØ╝ļŖö ņ┤Ø 7Ļ░£ņØś ņ¦łļ¼ĖņØä ĒĢ£ĻĖĆļĪ£ ņĀ£ņ×æĒĢśņśĆĻ│Ā, ņ¦łļ¼Ė ļŗ╣ 0ņŚÉņä£ 5ņĀÉ ņé¼ņØ┤ņŚÉņä£ 1ņĀÉ ļŗ©ņ£äļĪ£ ĻĘĖ ņĀĢļÅäņŚÉ ļö░ļØ╝ ņĀÉņłśļź╝ ļ¦żĻĖ░ļÅäļĪØ ĒĢśņśĆļŗż(Fig. 1). EDSQ ņäżļ¼Ėņ¦Ć ņżæ 2-5 ņśüņŚŁņØś ņ¦łļ¼ĖļōżņØĆ ĒÖśņ×ÉĻ░Ć ļŖÉļü╝ļŖö ņ¦łļ│æ ņ╣śļŻīņŚÉ ļīĆĒĢ£ ņČöņāüņĀüņØĖ ņāØĻ░üĻ│╝ ļ»┐ņØī, ņØśņé¼-ĒÖśņ×É Ļ┤ĆĻ│äņØś ļ¦īņĪ▒ļÅä, ņĢłņĢĮ ņé¼ņÜ®ņØś ĒÄĖļ”¼ĒĢ© ļō▒ņØä ĒÅēĻ░ĆĒĢśĻĖ░ ļĢīļ¼ĖņŚÉ ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ļŖö ĒżĒĢ©ĒĢśņ¦Ć ņĢŖņĢśļŗż. ņäżļ¼Ėņ¦ĆļŖö ņÖĖļל ļīĆĻĖ░ Ļ│ĄĻ░äņŚÉņä£ ĒÖśņ×ÉĻ░Ć ņ¦üņĀæ ņČ£ļĀźļ¼╝ņØä ņØĮņ¢┤ļ│┤ļ®┤ņä£ ņ▓┤Ēü¼ĒĢśļŖö ļ░®ņŗØņ£╝ļĪ£ ņ×æņä▒ĒĢśņśĆļŗż.
ņĀäņ▓┤ ļīĆņāü ĒÖśņ×ÉĻĄ░ ļé┤ņŚÉņä£ 2Ļ░£ņØś ņĮöĒśĖĒŖĖļź╝ ļ│äĻ░£ļĪ£ ņäżņĀĢĒĢ£ Ēøä ņĮöĒśĖĒŖĖ 1ņØä ļīĆņāüņ£╝ļĪ£ļŖö 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ņØś ņ┤łĻĖ░ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅä ņäżļ¼Ė ņĀÉņłśņŚÉ ņśüĒ¢źņØä ļ»Ėņ╣śļŖö ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÅēĻ░Ć ĒĢŁļ¬®ņŚÉ ļīĆĒĢ£ ļČäņäØņØä ņŗ£Ē¢ēĒĢśņśĆĻ│Ā, ņĮöĒśĖĒŖĖ 2ļź╝ ļīĆņāüņ£╝ļĪ£ļŖö 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ ņĀÉņĢł ļłäņĀü ĻĖ░Ļ░äņŚÉ ļö░ļźĖ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅä ņäżļ¼Ė ņĀÉņłśņØś ņČöņØ┤ ļČäņäØņØä ņŗ£Ē¢ēĒĢśņśĆļŗż.
0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ņØś ņĀÉņĢł Ēøä ņ┤łĻĖ░ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅä ĒÅēĻ░Ćļź╝ ņ£äĒĢ┤ ņĀäņ▓┤ ļīĆņāü ĒÖśņ×É ņżæ ņĀÉņĢł ņŗ£ņ×æ Ēøä 3Ļ░£ņøö ņØ┤ļé┤ņŚÉ ņ▓½ ņäżļ¼Ė ĒÅēĻ░Ćļź╝ ņŗ£Ē¢ēĒĢ£ ĒÖśņ×ÉļōżņØä ņĮöĒśĖĒŖĖ 1ļĪ£ ņäżņĀĢĒĢśņśĆļŗż. ņäżļ¼Ėņ¦Ć ļé┤ 7Ļ░£ ĒĢŁļ¬®ņØś Ļ░üĻ░üņØś ņĀÉņłśņÖĆ ņŚ░ļĀ╣, ņä▒ļ│ä, ņ▓½ ņäżļ¼Ė ĒÅēĻ░Ć ņŗ£ņØś ņĀÉņĢł ĻĖ░Ļ░ä(Ļ░£ņøö), ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÅēĻ░Ć ĒĢŁļ¬®ņ£╝ļĪ£ņä£ ļ¦łņØ┤ļ┤äņāśņØś ņĢĢņČ£ļŖź(expressibility), ļ¦łņØ┤ļ┤äņØś Ēś╝ĒāüļÅä(turbidity), ļłłļ¼╝ ļČäļ╣äļ¤ē, Ļ░üļ¦ēņ¦ĆĻ░ü ņŚŁņ╣ś, Ļ░üļ¦ē ļ»Ėļ×Ć, ņĢłĻĄ¼Ēæ£ļ®┤ņŚ╝ņāēņ¦Ćņłś(ocular staining score, OSS), ļłłļ¼╝ ļé┤ matrix metalloproteinase 9 (MMP-9) ļ░£Ēśä ļō▒ĻĖē ļ░Å ņĢłĻĄ¼Ēæ£ļ®┤ņ¦łĒÖśņ¦Ćņłś(ocular surface disease index, OSDI) ņäżļ¼Ė ņĀÉņłśļź╝ ĒżĒĢ©ĒĢ£ ņ┤Ø 11Ļ░£ ĒĢŁļ¬® Ļ░ä ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļČäņäØĒĢśņśĆļŗż. ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÅēĻ░Ć ĒĢŁļ¬®ļōżņØĆ ņäżļ¼Ė ĒÅēĻ░Ć ņŗ£Ē¢ēņØ╝ ĒżĒĢ© Ļ│╝Ļ▒░ 2ņŻ╝ ņØ┤ļé┤ņŚÉ ņŗ£Ē¢ēĒĢ£ Ļ▓Ćņé¼ Ļ▓░Ļ│╝ļ¦īņØä ļČäņäØ ļīĆņāüņ£╝ļĪ£ ĒĢśņśĆļŗż.
0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ņØś ņĀÉņĢł ļłäņĀü ĻĖ░Ļ░äņŚÉ ļö░ļźĖ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅä ļ│ĆĒÖö ņŚ¼ļČĆļź╝ ĒÖĢņØĖĒĢśĻĖ░ ņ£äĒĢ┤, ņĀäņ▓┤ ļīĆņāü ĒÖśņ×É ņżæ ņĄ£ņåī 2Ļ░£ņøö ņØ┤ņāü Ļ░äĻ▓®ņ£╝ļĪ£ 2ĒÜī ņØ┤ņāüņØś ņäżļ¼ĖņØä ņŗ£Ē¢ēĒĢ£ ņĀüņØ┤ ņ׳ļŖö ĒÖśņ×ÉļōżņØä ņĮöĒśĖĒŖĖ 2ļĪ£ ņäżņĀĢĒĢśņśĆļŗż. ņĮöĒśĖĒŖĖ 2 ļé┤ņŚÉņä£ ņĄ£ņ┤ł ņäżļ¼ĖĻ│╝ ņØ┤Ēøä ņĄ£ņåī 2Ļ░£ņøö ņØ┤ņāüņØś Ļ░äĻ▓®ņØś ņŚ░ ņØ┤ņØĆ 2ĒÜīņØś ņČöĻ░Ć ņäżļ¼ĖņØä ļīĆņāüņ£╝ļĪ£ ĒĢśņŚ¼, ņ┤Ø 3ĒÜīņØś ņäżļ¼Ėņ¦ĆņŚÉņä£ 7Ļ░£ ĒĢŁļ¬® Ļ░üĻ░üņØś ņĀÉņłśņØś ņŗ£Ļ░äņŚÉ ļö░ļźĖ ļ│ĆĒÖöļź╝ ļČäņäØĒĢśņśĆļŗż.
ļ¦łņØ┤ļ┤äņāśņØś ņĢĢņČ£ļŖźņØĆ ļ®┤ļ┤ēņ£╝ļĪ£ ņ£äļłłĻ║╝ĒÆĆĒīÉņØä Ļ░Ćļ│ŹĻ▓ī ļłīļĀĆņØä ļĢī ņżæņŗ¼ 5Ļ░£ņØś ļ¦łņØ┤ļ┤äņāś ņżæņŚÉņä£ ņĢĢņČ£ Ļ░ĆļŖźĒĢ£ ļ¦łņØ┤ļ┤äņāśņØś Ļ░£ņłśņŚÉ ļö░ļØ╝ņä£, 5Ļ░£ļŖö 0ļō▒ĻĖē, 3-4Ļ░£ļŖö 1ļō▒ĻĖē, 1-2Ļ░£ļŖö 2ļō▒ĻĖē, 0Ļ░£ļŖö 3ļō▒ĻĖēņ£╝ļĪ£ ļČäļźśĒĢśņśĆļŗż[21]. ļ¦łņØ┤ļ┤äņāś ļČäļ╣äļ¼╝ņØś Ēś╝ĒāüļÅäļŖö ņĢäļל ļłłĻ║╝ĒÆĆĒīÉ ņżæņŗ¼ņØä ņåÉĻ░ĆļØĮ ļüØņ£╝ļĪ£ ļłīļĀĆņØä ļĢī ņżæņŗ¼ļČĆ 8Ļ░£ņØś Ļ░£ĻĄ¼ļČĆņŚÉņä£ ļČäļ╣äļÉśļŖö ļ¦łņØ┤ļ┤äņØä Ļ┤Ćņ░░ĒĢśņŚ¼ Ēł¼ļ¬ģĒĢśļ®┤ 0ļō▒ĻĖē, ļŗ©ņł£ Ēś╝ĒāüĒĢśļ®┤(cloudy) 1ļō▒ĻĖē, ļģĖĒÅÉļ¼╝ Ļ░ĆļŻ©ļź╝ ļÅÖļ░śĒĢ£ Ēś╝Ēāü ņŗ£(cloudy with debris) 2ļō▒ĻĖē, ļæÉĻ╗ŹĻ│Ā ņ╣śņĢĮ Ļ░ÖņØ┤ ļ░░ņČ£ļÉśļ®┤ 3ļō▒ĻĖēņ£╝ļĪ£ ļČäļźśĒĢśņśĆļŗż[22]. ņē¼ļź┤ļ©ĖĻ▓Ćņé¼ļŖö ņĀÉņĢłļ¦łņĘ© ņŚåņØ┤ Ēæ£ņżĆ Ļ▓Ćņé¼ņ¦Ć(Eagle Vision, Memphis, TN, USA)ļź╝ ņĢäļלļłłĻ║╝ĒÆĆ ļ░öĻ╣ź 1/3 ņ¦ĆņĀÉņŚÉņä£ ņĢäļל ļłłĻ║╝ĒÆĆļéŁņŚÉ 5ļČäĻ░ä ņ£äņ╣śņŗ£Ēé© Ēøä ļłłļ¼╝ņØ┤ ņĀ¢ņØĆ ĻĖĖņØ┤(mm)ļź╝ ņĖĪņĀĢĒĢśņśĆļŗż. ļłłļ¼╝ ļé┤ MMP-9 ņĖĪņĀĢņØĆ InflammaDry┬« (Quidel, San Diego, CA, USA)ņØä ņé¼ņÜ®ĒĢśņśĆļŗż. ņĀÉņĢłļ¦łņĘ© ņŚåņØ┤ ņĢäļל ļłłĻ║╝ĒÆĆ Ļ▓░ļ¦ēņØś ņŚ¼ļ¤¼ ņ£äņ╣śņŚÉ 6-8ĒÜī ņØ┤ņāü ņāśĒöī ņłśņ¦æĻĖ░ ļüØņØä ņĀæņ┤ēņŗ£ņ╝£ ļłłļ¼╝ņØä ĒØĪņłśņŗ£Ēé© Ēøä Ļ▓Ćņé¼ ņ╣┤ņäĖĒŖĖņŚÉ Ļ▓░ĒĢ®ņŗ£ĒéżĻ│Ā, ņÖäņČ®ņĢĪņŚÉ Ēīü ļüØņØä 20ņ┤ł ņØ┤ņāü ļŗ┤ĻĘĖĻ│Ā 10ļČä ĒøäņŚÉ Ļ▓Ćņé¼ņ░ĮņŚÉ ļéśĒāĆļéśļŖö ļ╣©Ļ░äņāēņØś ņäĀņØś ņ¦äĒĢ£ ņĀĢļÅäņŚÉ ļö░ļØ╝ ņØīņä▒(0ļō▒ĻĖē), trace (1ļō▒ĻĖē), ņĢĮņ¢æņä▒(2ļō▒ĻĖē), ņ¢æņä▒(3ļō▒ĻĖē), Ļ░Ģņ¢æņä▒(4ļō▒ĻĖē)ņØś 5ļŗ©Ļ│äņØś ļō▒ĻĖēņ£╝ļĪ£ ĒīÉļŗ©ĒĢśņśĆļŗż[23]. Ļ░üļ¦ēņ¦ĆĻ░üļŖźņØĆ Cochet-Bonnet Ļ░üļ¦ēņ¦ĆĻ░üĻ│ä(Luneau ophthalmology, Chartres Cedex, France)ļź╝ ņØ┤ņÜ®ĒĢśņŚ¼ Ļ░üļ¦ē ņżæņŗ¼ļČĆņŚÉņä£ ĒÅēĻ░ĆĒĢśņśĆņ£╝ļ®░, ļéśņØ╝ļĪĀņé¼ļź╝ Ļ░üļ¦ēļ®┤ņŚÉ ņłśņ¦üņ£╝ļĪ£ ņĄ£ņåīĒĢ£ņØś ĻĄ¼ļČĆļ¤¼ņ¦ÉņØ┤ ņāØĻĖĖ ļĢīĻ╣īņ¦Ć ņĢĢļ░ĢĒĢ┤ ņĄ£ņ┤łļĪ£ ņ¢æņä▒ ļ░śņØæņØä ļéśĒāĆļé╝ ļĢīņØś ļéśņØ╝ļĪĀņé¼ņØś ĻĖĖņØ┤ļź╝ Ļ░üļ¦ēņ¦ĆĻ░ü ņŚŁņ╣śĻ░Æ(mm)ņ£╝ļĪ£ Ēæ£ņŗ£ĒĢśņśĆļŗż. Ļ░üļ¦ēļ»Ėļ×ĆņØś ņĀĢļÅäļŖö ņāØļ”¼ņŗØņŚ╝ņłśļź╝ ļ¼╗Ē×ī ĒöīļŻ©ņśżļĀłņäĖņØĖ ņóģņØ┤(Haag-Streit international, Koniz, Switzerland)ļź╝ ņØ┤ņÜ®ĒĢ£ Ļ░üļ¦ēĒśĢĻ┤æņŚ╝ņāē ņĀÉņłśļĪ£ ĒīÉļŗ©ĒĢśņśĆņ£╝ļ®░, ļ»ĖĻĄŁĻĄŁļ”ĮņĢłņŚ░ĻĄ¼ņåī(The National Eye Institute/Industry, Scale) ĻĖ░ņżĆņ£╝ļĪ£ Ļ░üļ¦ēņØä 5ļō▒ļČäĒĢśņŚ¼ Ļ░üĻ░ü 0-3ņĀÉņ£╝ļĪ£ ņĀÉņłśļź╝ ļČĆņŚ¼ĒĢśĻ│Ā ņ┤Ø 15ņĀÉņ£╝ļĪ£ Ļ░üļ¦ēļ»Ėļ×ĆņØś ņżæņ”ØļÅäļź╝ ĒÅēĻ░ĆĒĢśņśĆļŗż[24]. ņĢłĻĄ¼Ēæ£ļ®┤ņŚ╝ņāēņ¦ĆņłśļŖö Sj├ČgrenŌĆÖs International Collaborative Clinical Alliance (SICCA) OSSļź╝ ĻĖ░ņżĆņ£╝ļĪ£ Ļ░üļ¦ēĻ│╝ Ļ▓░ļ¦ēņØś ņĀÉņłśļź╝ ĒĢ®ņé░ĒĢśņŚ¼ Ēæ£ĻĖ░ĒĢśņśĆļŗż[25].
ņĮöĒśĖĒŖĖ 1ņŚÉņä£ ņŚ░ļĀ╣, ņ▓½ ņäżļ¼Ė ĒÅēĻ░Ć ņŗ£ņØś ņĀÉņĢł ĻĖ░Ļ░ä ļ░Å ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÅēĻ░Ć ĒĢŁļ¬® Ļ▓░Ļ│╝ņÖĆ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅä ņäżļ¼Ė ņĀÉņłś Ļ░ä ņāüĻ┤ĆĻ┤ĆĻ│äļŖö SpearmanŌĆÖs rank ņāüĻ┤Ć ļČäņäØņØä ņØ┤ņÜ®ĒĢśņśĆņ£╝ļ®░, ņä▒ļ│äņŚÉ ļö░ļźĖ ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅä ņäżļ¼Ė ņĀÉņłśņØś ņ░©ņØ┤ļŖö Mann-Whitney U ļČäņäØņØä ņØ┤ņÜ®ĒĢśņśĆļŗż. ņĮöĒśĖĒŖĖ 2ņŚÉņä£ ņĀÉņĢł ļłäņĀü ĻĖ░Ļ░äņŚÉ ļö░ļźĖ ņäżļ¼Ė ņĀÉņłśņØś ņČöņäĖņĀü ļ│ĆĒÖöļź╝ ļČäņäØĒĢśĻĖ░ ņ£äĒĢ┤ Ļ░ü ņäżļ¼Ė ĒÜīņ░© Ļ░äņØś ņĀÉņłśļź╝ ņ¦Øņ¦Ćņ¢┤ Wilcoxon signed rank ļČäņäØņØä ņŗ£Ē¢ēĒĢśņśĆļŗż. ĒåĄĻ│äņĀü ņ▓śļ”¼ļŖö SPSS for Window (SPSS Version 26.0 Inc., IBM Corp., Armonk, NY, USA) ĒåĄĻ│ä ĒöäļĪ£ĻĘĖļשņØä ņØ┤ņÜ®ĒĢśņśĆĻ│Ā ņ£ĀņØś ĒÖĢļźĀ(p-value)ņØ┤ 0.05 ļ»Ėļ¦īņØĖ Ļ▓ĮņÜ░ļź╝ ĒåĄĻ│äņĀüņ£╝ļĪ£ ņ£ĀņØśĒĢ£ Ļ▓āņ£╝ļĪ£ Ļ░äņŻ╝ĒĢśņśĆļŗż. Ļ▓░Ļ│╝Ļ░ÆļōżņØĆ ĒÅēĻĘĀ ┬▒ Ēæ£ņżĆĒÄĖņ░©ņØś ĒśĢņŗØņ£╝ļĪ£ Ēæ£ņŗ£ĒĢśņśĆļŗż.
ņĮöĒśĖĒŖĖ 1ņŚÉļŖö ņ┤Ø 39ļ¬ģ 74ņĢłņØ┤ ĒżĒĢ©ļÉśņŚłņ£╝ļ®░ ĒĢ┤ļŗ╣ ņØĖĻĄ¼ĒĢÖņĀü ļČäĒżņÖĆ 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ņØś ņĀÉņĢł ņŗ£ņ×æ 3Ļ░£ņøö ņØ┤ļé┤ņŚÉ ņ▓śņØī ņŗ£Ē¢ēĒĢ£ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÅēĻ░Ć ĒĢŁļ¬®ļōżņØĆ Table 1ņŚÉ ļéśĒāĆļé┤ņŚłļŗż. ņĮöĒśĖĒŖĖ 2ļŖö ņ┤Ø 34ļ¬ģņØä ĒżĒĢ©ĒĢśņśĆĻ│Ā ĒĢ┤ļŗ╣ ņØĖĻĄ¼ĒĢÖņĀü ļČäĒżļŖö Table 2ņŚÉ ļéśĒāĆļé┤ņŚłļŗż.
ņ╣śļŻīņŚÉ ļīĆĒĢ£ Ļ▒▒ņĀĢ ĒÅēĻ░Ć ņśüņŚŁņØś 4Ļ░Ćņ¦Ć ņ¦łļ¼Ė: ņ¦łļ¼Ė 1) ņĀÉņĢł ņ¦üĒøä ļłłņØ┤ ļČłĒÄĖĒ¢łļŖöņ¦ĆņØś ņĀÉņłśļŖö ņŚ¼ņä▒ņŚÉņä£ ņ£ĀņØśĒĢśĻ▓ī ļŹö ļåÆņĢśĻ│Ā(p=0.048), ņ¦łļ¼Ė 2) ņĀÉņĢłĻ░ÉņØ┤ ļČłĒÄĖĒĢĀĻ╣īļ┤É ļ»Ėļ”¼ Ļ▒▒ņĀĢņØ┤ ļÉśņŚłļŖöņ¦ĆņØś ņĀÉņłśļŖö ņĢłņĢĮ ņĀÉņĢł ĻĖ░Ļ░äĻ│╝ ņ£ĀņØśĒĢ£ ņØīņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝(r=-0.389, p=0.002), ļłłļ¼╝ ļČäļ╣äļ¤ēĻ│╝ļŖö ņ£ĀņØśĒĢ£ ņ¢æņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņśĆņ£╝ļ®░(r=0.289, p=0.042), ņ¦łļ¼Ė 3) ņĀÉņĢł ņ¦üĒøä ļéśļÅä ļ¬©ļź┤Ļ▓ī ļłłņØ┤ Ļ╣£ļ╣ĪņśĆļŖöņ¦ĆņØś ņĀÉņłśļŖö Ļ░üļ¦ēĒśĢĻ┤æņŚ╝ņāē ņĀÉņłśņÖĆ ņ£ĀņØśĒĢ£ ņ¢æņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņśĆļŗż(r=0.297, p=0.015). ņ¦łļ¼Ė 4) ņĀÉņĢł ļČłĒÄĖĻ░ÉņØ┤ ņŗ¼ņĀü ļČĆļŗ┤ņØ┤ ļÉśņŚłļŖöņ¦ĆņØś ņĀÉņłśļŖö ņŚ░ļĀ╣Ļ│╝ ņØīņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝(r=-0.247, p=0.049), ņĢłĻĄ¼Ēæ£ļ®┤ņŚ╝ņāēņ¦Ćņłś(r=0.298, p=0.047) ļ░Å ļłłļ¼╝ ļé┤ MMP-9 ļ░£Ēśä ļō▒ĻĖēĻ│╝ļŖö ņ£ĀņØśĒĢ£ ņ¢æņØś ņāüĻ┤ĆĻ┤ĆĻ│ä(r=0.287, p=0.032)ļź╝ ļ│┤ņśĆļŗż(Table 3). ņ×ÉĻ░Ć ņł£ņØæļÅä ĒÅēĻ░Ć ņśüņŚŁņØś 3Ļ░Ćņ¦Ć ņ¦łļ¼Ė ņżæ ņ¦łļ¼Ė 5) ņ¦Ćļé£ 4ņŻ╝Ļ░ä ļéśļ”ä ņל ņĀÉņĢłĒ¢łļŗżĻ│Ā ņāØĻ░üĒĢśļŖöņ¦ĆņØś ņĀÉņłśļŖö ļłłļ¼╝ ļé┤ MMP-9 ļ░£Ēśä ļō▒ĻĖēĻ│╝ ņ£ĀņØśĒĢ£ ņØīņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņśĆņ£╝ļ®░(r=-0.320, p=0.016), ņ¦łļ¼Ė 6) ņĀÉņĢłĒĢśļŖö Ļ▓āņØä ņל ņ×Ŗņ¢┤ļ▓äļĀĖļŖöņ¦ĆņØś ņĀÉņłśņÖĆ ņ¦łļ¼Ė 7) ņØ╝ļČĆļ¤¼ ņĀÉņĢłņØä ņĢłĒĢśĻĖ░ļÅä Ē¢łļŖöņ¦ĆņØś ņĀÉņłśņÖĆ ņ£ĀņØśĒĢ£ ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņØĖ ņØĖņ×ÉļŖö ņŚåņŚłļŗż(Table 4).
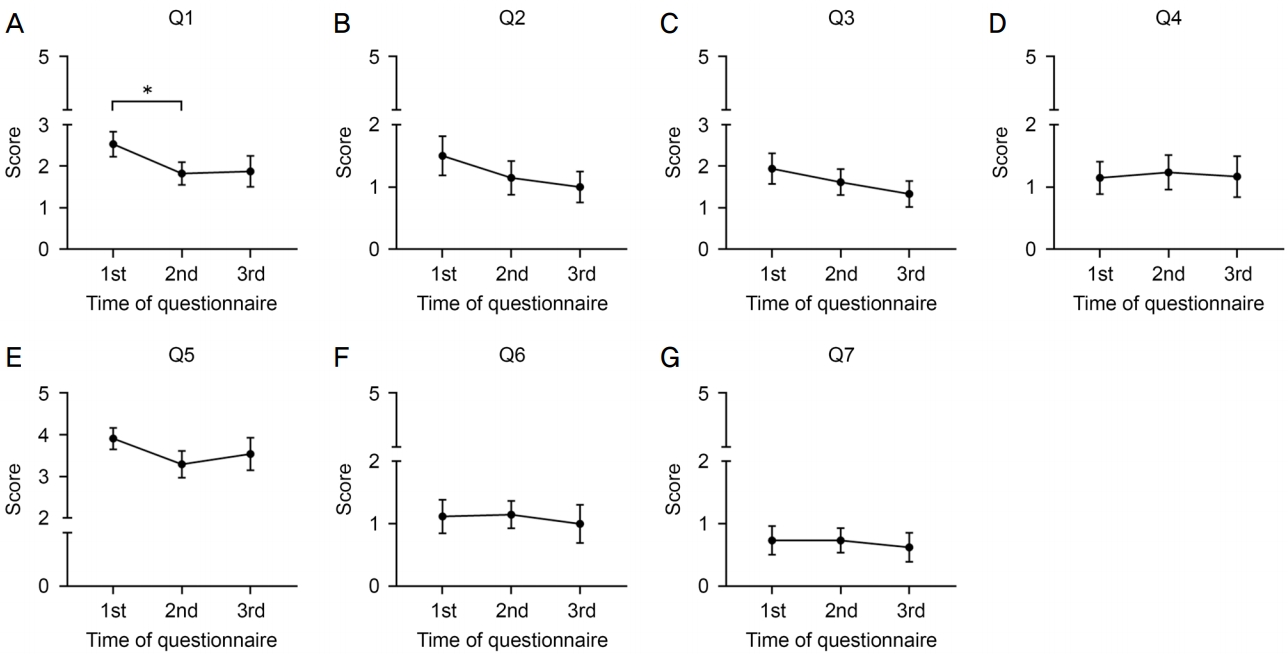
ņĄ£ņåī 2Ļ░£ņøö Ļ░äĻ▓®ņØś ņŚ░ņØ┤ņØĆ 3ĒÜīņØś ņäżļ¼Ė ņżæ ņ▓½ ļ▓łņ¦Ė ņäżļ¼Ė ņŗ£Ļ╣īņ¦ĆņØś ņĀÉņĢł ĻĖ░Ļ░äņØĆ 3.56 ┬▒ 2.80Ļ░£ņøö, ļæÉ ļ▓łņ¦Ė ņäżļ¼Ė ņŗ£Ļ╣īņ¦ĆļŖö 6.21 ┬▒ 3.31Ļ░£ņøö, ĻĘĖļ”¼Ļ│Ā ņäĖ ļ▓łņ¦Ė ņäżļ¼Ė ņŗ£Ļ╣īņ¦ĆļŖö 8.83 ┬▒ 2.87Ļ░£ņøöņØ┤ņŚłļŗż. ņ╣śļŻīņŚÉ ļīĆĒĢ£ Ļ▒▒ņĀĢ ĒÅēĻ░Ć ņśüņŚŁ ņżæ ņ¦łļ¼Ė 1-3ņØś Ļ░ü ĒÅēĻĘĀ ņĀÉņłśļōżņØĆ ļłäņĀü ņĀÉņĢł ĻĖ░Ļ░äņØ┤ ĻĖĖņ¢┤ņ¦łņłśļĪØ Ļ░ÉņåīĒĢśļŖö Ļ▓ĮĒ¢źņØä ļ│┤ņśĆĻ│Ā(Fig. 2A-C), ĻĘĖņżæ ĒŖ╣Ē׳, ņ¦łļ¼Ė 1) ņĀÉņĢł ņ¦üĒøä ļłłņØ┤ ļČłĒÄĖĒ¢łļŖöņ¦ĆņØś ņĀÉņłśļŖö 2ĒÜīņ░© ņäżļ¼Ė ņŗ£ ņĄ£ņ┤ł ņäżļ¼Ė ņŗ£ ļīĆļ╣ä ņ£ĀņØśĒĢśĻ▓ī Ļ░ÉņåīĒĢśņśĆļŗż(p=0.031, Fig. 2A). ņ¦łļ¼Ė 4-7ņØś ņĀÉņłśļŖö ļłäņĀü ņĀÉņĢł ĻĖ░Ļ░äņŚÉ ļö░ļØ╝ ļ│ĆĒĢśņ¦Ć ņĢŖņĢśļŗż(Fig. 2D-G).
ņĢłĻĄ¼ Ēæ£ļ®┤ ņ¦łĒÖśļōżņØś ņ╣śļŻīņŚÉ ņ׳ņ¢┤ ņØ╝ļ░śņĀüņ£╝ļĪ£ ņĢłņĢĮņĀ£ņØś ĻĄŁņåīņĀü Ēł¼ņĢĮņØ┤ ļ¦żņÜ░ Ēü░ ļ╣äņżæņØä ņ░©ņ¦ĆĒĢśļŖöļŹ░, ĒÖśņ×ÉņØś ņĢĮņĀ£ ņł£ņØæļÅäĻ░Ć ņä▒Ļ│ĄņĀüņØĖ ņ╣śļŻīņŚÉ ņ׳ņ¢┤ ļ¦żņÜ░ ņżæņÜöĒĢśļŗż. Ļ░üļ¦ēņØś ĻĄ┤ņĀłĻĄÉņĀĢņłśņłĀņØ┤ļéś ļ░▒ļé┤ņןņłśņłĀ ļō▒ņØś ņłśņłĀ Ēøä ņĢłņĢĮ ņé¼ņÜ®ņØĆ ļīĆĻ░£ ņĀÉņĢł ĻĖ░Ļ░äņØ┤ 1ņŻ╝-1ļŗ¼ ņĀĢļÅäļĪ£ ņ¦¦Ļ│Ā ļ¬ģĒÖĢĒĢ£ Ēł¼ņĢĮ ļ¬®ņĀüņØä ĒÖśņ×ÉĻ░Ć ņØ┤ļ»Ė ņØĖņ¦ĆĒĢśĻ│Ā ņ׳ņ£╝ļ®░, Ļ░ÉņŚ╝ņä▒ Ļ░üļ¦ēņŚ╝Ļ│╝ Ļ░ÖņØĆ ĻĖēņä▒ĻĖ░ ņ¦łĒÖśņŚÉņä£ļŖö ĒÜ©Ļ│╝ņĀüņØĖ ņĢłņĢĮ ņé¼ņÜ®ņØ┤ ļŗ©ĻĖ░Ļ░ä ļé┤ ĒÖśņ×ÉĻ░Ć ņ×ÉĻ░üĒĢĀ ņłś ņ׳ņØä ņĀĢļÅäļĪ£ ņ”ØņāüņØś ĒśĖņĀäņØä Ļ░ĆņĀĖņśżĻ│ż ĒĢśĻĖ░ ļĢīļ¼ĖņŚÉ ņĢĮņĀ£ ņé¼ņÜ®ņØś ņł£ņØæļÅäĻ░Ć ļīĆĻ░£ ņóŗņØĆ ĒÄĖņØ┤ļŗż. ļ░śļ®┤, ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø, ļģ╣ļé┤ņן ļō▒ņØś ņןĻĖ░Ļ░ä ĻŠĖņżĆĒĢ£ ņĢłņĢĮ ņĀÉņĢłņØä ĒĢäņÜöļĪ£ ĒĢśļŖö ļ¦īņä▒ ņ¦łĒÖśņŚÉņä£ļŖö ņāüļīĆņĀüņ£╝ļĪ£ ņĢłņĢĮņĀ£ņØś ņł£ņØæļÅäĻ░Ć ļ¢©ņ¢┤ņ¦äļŗż. ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÖśņ×ÉņŚÉņä£ ņĢłņĢĮņĀ£ņØś ļ╣äņł£ņØæ(non-compliance)ņØĆ ņŗ£ļĀźņĀĆĒĢś ļ┐Éļ¦ī ņĢäļŗłļØ╝, ņŚģļ¼┤ ņāØņé░ņä▒ņØś ņĀĆĒĢś ļ░Å ņéČņØś ņ¦ł ņĀĆĒĢś ļō▒ņØś ņé¼ĒÜīņĀü ļ¼ĖņĀ£ļĪ£ ņØ┤ņ¢┤ņ¦ł ņłś ņ׳ņ£╝ļ®░[26], ĻĘĖņżæ 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ ņĀÉņĢłņĀ£ņÖĆ Ļ░ÖņØ┤ ņŗżņĀ£ ņżæļō▒ļÅä ņØ┤ņāüņØś ņŗ¼ĒĢ£ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”ØņŚÉņä£ ņŻ╝ļĪ£ ņé¼ņÜ®ļÉśĻĖ░ ņĢłņĢĮņĀ£ņŚÉ ņ׳ņ¢┤ņä£ļŖö ĻŠĖņżĆĒĢ£ ņĀÉņĢłņØ┤ ņ╣śļŻī ĒÜ©Ļ│╝ ĒīÉļŗ© ļ░Å ņä▒Ļ│ĄņĀüņØĖ ņ╣śļŻīņŚÉ ņ׳ņ¢┤ ļ¦żņÜ░ ņżæņÜöĒĢśļŗż. ĻĘĖļ¤¼ļéś, 0.05% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ņØ┤ļéś ļ”¼Ēö╝ĒģīĻĘĖļØ╝ņŖżĒŖĖ(lifitegrast) ļō▒ņØś ņÖĖņĢłļČĆ ĒĢŁņŚ╝ņ”Ø ņĀÉņĢłņĀ£ ņé¼ņÜ® ĒÖśņ×É ņżæ ņĀÉņĢł ņŗ£ ĒåĄņ”Ø, Ļ░ĆļĀżņøĆ, ņ×æņŚ┤Ļ░É ļĢīļ¼ĖņŚÉ ņĢĮļ¼╝ņØä ņé¼ņÜ®ĒĢśņ¦Ć ņĢŖĻ▒░ļéś ņżæļŗ©ĒĢ£ ļ░ö ņ׳ņŚłļŗżĻ│Ā ņĢīļĀżņ¦ä ĒÖśņ×ÉļŖö 2.5-60%ņØś ļŗżņ¢æĒĢ£ ļ╣łļÅäļĪ£ ļ│┤Ļ│ĀļÉ£ Ļ▓āņ▓śļ¤╝[10,27,28], ņĢłņĢĮņĀ£ņØś ļ╣äņł£ņØæļÅäļŖö ĒÖśņ×Éļ¦łļŗż ņāüņØ┤ĒĢśļŗż. ņØ┤ņŚÉ ļ│Ė ņŚ░ĻĄ¼ļź╝ ĒåĄĒĢ┤ ĒĢŁņŚ╝ņ”Ø ĒÜ©Ļ│╝Ļ░Ć ņÜ░ņłśĒĢ£ ļ░śļ®┤, ņĀÉņĢł ņŗ£ ĒåĄņ”Ø ļ░Å ņ×ÉĻĘ╣Ļ░ÉņØś ļ░£ņāØņØ┤ ņĢīļĀżņ¦ä 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ņØś ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅäņŚÉ ļ»Ėņ╣śļŖö ņśüĒ¢źļĀź ņØĖņ×Éļź╝ ĒāÉņāēĒĢśņśĆļŗż. ĻĘĖ Ļ▓░Ļ│╝ ņŚ¼ņä▒Ļ│╝ ņāüļīĆņĀüņ£╝ļĪ£ ņĀŖņØĆ ĒÖśņ×ÉņŚÉņä£, Ļ░üļ¦ēļ»Ėļ×Ć, ņĢłĻĄ¼Ēæ£ļ®┤ņŚ╝ņāēņ¦Ćņłś, ļłłļ¼╝ ļé┤ MMP-9 ļ░£Ēśä ņĀĢļÅä, ļłłļ¼╝ ļČäļ╣äļ¤ēņØ┤ ļ│┤ļŗż ļåÆņØĆ ĒÖśņ×ÉņŚÉņä£, ĻĘĖļ”¼Ļ│Ā ņĀÉņĢł ĻĖ░Ļ░äņØ┤ ņāüļīĆņĀüņ£╝ļĪ£ ņ¦¦ņØĆ ĒÖśņ×ÉņŚÉņä£ ņĀÉņĢł ņŗ£ ļČłĒÄĖĻ░ÉņØä ļŹö Ēü¼Ļ▓ī ĒśĖņåīĒĢ£ ļ░śļ®┤, ņĀÉņĢł ļłäņĀü ĻĖ░Ļ░äņØ┤ ņ”ØĻ░ĆĒĢĀņłśļĪØ ņĀÉņĢł ņŗ£ ļČłĒÄĖĻ░ÉņØĆ Ļ░ÉņåīĒĢśņśĆņ£╝ļ®░, ļłłļ¼╝ ļé┤ MMP-9 ļ░£Ēśä ņĀĢļÅäĻ░Ć ļåÆņØäņłśļĪØ ņĀÉņĢł ņł£ņØæļÅäĻ░Ć ļ¢©ņ¢┤ņ¦äļŗżļŖö Ļ▓░Ļ│╝ļź╝ ņ¢╗ņŚłļŗż.
ņĮöĒśĖĒŖĖ 1ņŚÉņä£ ņ╣śļŻīņŚÉ ļīĆĒĢ£ Ļ▒▒ņĀĢ ĒÅēĻ░Ć ņśüņŚŁņØś 4Ļ░Ćņ¦Ć ņ¦łļ¼Ė ņżæ ņ¦łļ¼Ė 1) ņĀÉņĢł ņ¦üĒøä ļłłņØ┤ ļČłĒÄĖĒ¢łļŖöņ¦ĆņØś ņĀÉņłśļŖö ņŚ¼ņä▒ņŚÉņä£ ļŹö ļåÆĻ▓ī ļéśĒāĆļé¼ļŗż. Ļ│╝Ļ▒░ ĒĢ£ ņŚ░ĻĄ¼ņŚÉ ļö░ļź┤ļ®┤ ņĢłĻĄ¼ ĒåĄņ”Ø ņ×Éņ▓┤ļŖö ņä▒ļ│äņØ┤ ņĢäļŗī OSDIņÖĆ Ļ░ÖņØĆ ņżæņ”ØļÅä ņ¦ĆĒæ£ņÖĆ ņ£ĀņØśĒĢ£ ņāüĻ┤Ćņä▒ņØä ļ│┤ņØĖļŗżĻ│Ā ņĢīļĀżņ¦ä ļ░ö ņ׳ļŖöļŹ░[29], ļ╣äļĪØ ļ│Ė ņŚ░ĻĄ¼ņØś ņĮöĒśĖĒŖĖ 1ņŚÉņä£ ļé©ņä▒Ļ│╝ ņŚ¼ņä▒ Ļ░ä OSDI ņĀÉņłśņØś ņ£ĀņØśĒĢ£ ņ░©ņØ┤ļŖö ņŚåņŚłņ¦Ćļ¦ī, ņŚ¼ņä▒ņŚÉņä£ ņŚ░ļĀ╣ņØ┤ ļŹö ļåÆņĢśĻ│Ā Ļ░üļ¦ēļ»Ėļ×ĆņØ┤ ļŹö ņŗ¼Ē¢łņ£╝ļ®░, ļłłļ¼╝ ļČäļ╣äļ¤ēņØ┤ 5.53 ┬▒ 3.72 mmļĪ£ ļé©ņä▒ņŚÉ ļ╣äĒĢ┤ Ēø©ņö¼ ļŹö ļé«ņĢśļŹś ņĀÉņØ┤(data not shown) ņ¦łļ¼Ė 1ņØś ņä▒ļ│ä Ļ░ä ņĀÉņłś ņ░©ņØ┤ļź╝ ņ£Āļ░£ĒĢśņśĆņØä Ļ▓āņØ┤ļØ╝ ņČöņĀĢļÉ£ļŗż. ļśÉĒĢ£, ļé©ļģĆ Ļ░ä ņĀÉņłś ņ░©ņØ┤ņØś ņ£ĀņØśņä▒ņØ┤ Ļ▓ĮĻ│äņä▒(p=0.048)ņØ┤ņŚłļŹś ņĀÉņØĆ ņ¦łļ¼Ė 1 ņĀÉņłśņØś ļé©ļģĆ ņ░©ņØ┤Ļ░Ć ņĀüņØĆ Ēæ£ļ│Ė ņłś ļĢīļ¼ĖņØ┤ņŚłņØä Ļ░ĆļŖźņä▒ ņŚŁņŗ£ ņŗ£ņé¼ĒĢ£ļŗż.
ņĮöĒśĖĒŖĖ 1ņŚÉņä£ ņ¦łļ¼Ė 2) ņĀÉņĢłĻ░ÉņØ┤ ļČłĒÄĖĒĢĀĻ╣īļ┤É ļ»Ėļ”¼ Ļ▒▒ņĀĢņØ┤ ļÉśņŚłļŖöņ¦ĆņØś ņĀÉņłśļŖö ņĀÉņĢł ĻĖ░Ļ░äņØ┤ ĻĖĖņłśļĪØ ļé«ņĢśļŖöļŹ░ ņØ┤ļŖö ņĀÉņĢł ņŗ£ ļČłĒÄĖĻ░ÉņŚÉ ļīĆĒĢ£ ĒĢÖņŖĄ ĒÜ©Ļ│╝ ļĢīļ¼ĖņØ┤ļØ╝Ļ│Ā ĒīÉļŗ©ļÉśļ®░, ņĮöĒśĖĒŖĖ 2ņŚÉņä£ ņ¦łļ¼Ė 1ņØś ņĀÉņłśĻ░Ć ņĄ£ņåī 2Ļ░£ņøö ņØ┤ņāü ņĀÉņĢłĒĢśņśĆņØä ņŗ£ ņ£ĀņØśĒĢśĻ▓ī ļé«ņĢäņ¦ä ņĀÉ ņŚŁņŗ£ ņØ┤ņÖĆ ņāüĒåĄĒĢ£ļŗż. ļłłļ¼╝ ļČäļ╣äļŖö Ļ░üļ¦ēņØś ĻĄ¼ņŗ¼ņä▒ ņ¦ĆĻ░ü ņŗĀĻ▓ĮņØś ņ×ÉĻĘ╣ņŚÉņä£ ņŗ£ņ×æĒĢ┤ņä£ ļłłļ¼╝ņāśņŚÉņä£ ļłłļ¼╝ņØ┤ ļČäļ╣äļÉśļŖö ļłłļ¼╝ ĻĖ░ļŖź ļŗ©ņ£ä(lacrimal functional unit)ņŚÉ ņØśĒĢ┤ ņĀĢņāüņĀüņ£╝ļĪ£ ņĪ░ņĀłļÉśļŖöļŹ░[30], ņĮöĒśĖĒŖĖ 1ņŚÉņä£ ņ¦łļ¼Ė 2ņØś ņĀÉņłśĻ░Ć ļłłļ¼╝ ļČäļ╣äļ¤ēĻ│╝ ņ¢æņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņØĖ ņĀÉņØĆ Ļ░üļ¦ēņØś ņ¦ĆĻ░ü ņŗĀĻ▓ĮņØ┤ ļ╣äĻĄÉņĀü ņĀĢņāüņĀüņØĖ ĒÖśņ×ÉņŚÉņä£ ļČłĒÄĖĻ░É ņŚŁņŗ£ ļŹö ņל ļŖÉļéä ņłś ņ׳ļŗżļŖö ņĀÉņØä ņČöņĀĢĒĢśĻ▓ī ĒĢ£ļŗż. ņĮöĒśĖĒŖĖ 1ņŚÉņä£ ņ¦łļ¼Ė 3) ņĀÉņĢł ņ¦üĒøä ļéśļÅä ļ¬©ļź┤Ļ▓ī ļłłņØ┤ Ļ╣£ļ╣ĪņśĆļŖöņ¦ĆņØś ņĀÉņłśĻ░Ć Ļ░üļ¦ēļ»Ėļ×Ć ņĀĢļÅäņÖĆ ņ£ĀņØśĒĢ£ ņ¢æņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņØĖ ņĀÉņØĆ ņĢäļ¦łļÅä Ļ░üļ¦ēļ»Ėļ×Ćņ£╝ļĪ£ ņØĖĒĢ┤ ĒÅēņāü ņŗ£ Ļ░üļ¦ēņØś ņ¦ĆĻ░ü ņŗĀĻ▓ĮņØ┤ ņēĮĻ▓ī ņ×ÉĻĘ╣ļÉĀ ņłś ņ׳ļŖö ĒÖśĻ▓ĮņŚÉņä£ ļéśĒāĆļéśļŖö ĒÖ£ņä▒ĒÖöļÉ£ ļłłĻ╣£ļ╣Īņ×ä ļ░śņé¼ ļĢīļ¼ĖņØ╝ Ļ▓āņØ┤ļŗż. ņ¦łļ¼Ė 4) ņĀÉņĢł ļČłĒÄĖĻ░ÉņØ┤ ņŗ¼ņĀü ļČĆļŗ┤ņØ┤ ļÉśņŚłļŖöņ¦ĆņØś ņĀÉņłśĻ░Ć Ļ│ĀļĀ╣ņØ╝ņłśļĪØ ņ£ĀņØśĒĢśĻ▓ī ļé«ņĢśļŹś Ļ▓░Ļ│╝ļŖö ĒåĄņāüņĀüņ£╝ļĪ£ ņŚ░ļĀ╣ņØ┤ ņ”ØĻ░ĆĒĢĀņłśļĪØ Ļ░üļ¦ēņØś ņ¦ĆĻ░üļŖźņØ┤ Ļ░ÉņåīĒĢ£ļŗżĻ│Ā ņĢīļĀżņ¦ä Ļ│╝Ļ▒░ ņŚ░ĻĄ¼ Ļ▓░Ļ│╝ļōżĻ│╝ ĻĘĖ ĻĖ░ņĀäņØä ņØ╝ļČĆ Ļ│Ąņ£ĀĒĢĀ Ļ▓āņØ┤ļØ╝ ĒīÉļŗ©ļÉ£ļŗż[31]. ļ░śļ®┤, ņĮöĒśĖĒŖĖ 1ņŚÉņä£ ņ¦łļ¼Ė 1-7ņØś ņĀÉņłś ļ¬©ļæÉ Ļ░üļ¦ēņ¦ĆĻ░ü ņŚŁņ╣śņÖĆ ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņØ┤ņ¦Ć ņĢŖņĢśļŖöļŹ░, ņØ┤ļŖö ņĮöĒśĖĒŖĖ 1ņŚÉņä£ņØś ĒÅēĻĘĀ Ļ░üļ¦ēņ¦ĆĻ░ü ņŚŁņ╣śĻ░ÆņØ┤ 5.24 ┬▒ 1.24 mmļĪ£ ļ╣äĻĄÉņĀü ņĀĢņāü ļ▓öņ£äņŚÉ Ļ░ĆĻ╣īņøĀĻĖ░ ļĢīļ¼ĖņØ┤ļØ╝Ļ│Ā ņāØĻ░üļÉśļ®░ ļ¦īņĢĮ ņŗĀĻ▓Įņśüņ¢æņä▒ Ļ░üļ¦ēļ│æņ”ØĻ│╝ Ļ░ÖņØ┤ Ļ░üļ¦ēņ¦ĆĻ░ü ņŚŁņ╣śĻ░Ć ļ¬ģļ░▒ĒĢśĻ▓ī Ļ░ÉņåīĒĢ£ ĒÖśņ×ÉļōżņØ┤ ļīĆĻ▒░ ĒżĒĢ©ļÉśņŚłļŹöļØ╝ļ®┤ ņäżļ¼Ė ņĀÉņłśļōżĻ│╝ Ļ░üļ¦ēņ¦ĆĻ░ü ņŚŁņ╣śĻ░Ć ņ£ĀņØśĒĢ£ ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņśĆņØä ņłśļÅä ņ׳Ļ▓Āļŗż. ĒĢśņ¦Ćļ¦ī, Ļ│ĀļĀ╣ņØ┤ļØ╝ļŖö ņĀÉ ņ×Éņ▓┤Ļ░Ć ļŗżņ¢æĒĢ£ ļŗżļźĖ ļ│æņĀü ņØĖņ×ÉļōżņØś ĻĄÉĒśĖ ņ×æņÜ®ņØś ņĪ┤ņ×¼ Ļ░ĆļŖźņä▒ņØä ņŗ£ņé¼ĒĢśĻĖ░ ļĢīļ¼ĖņŚÉ, ļéśņØ┤Ļ░Ć ļ¦ÄņØäņłśļĪØ ņĀÉņĢł ļČłĒÄĖĻ░ÉņØ┤ ļŹö ņĀüņØä Ļ▓āņØ┤ļØ╝Ļ│Ā ļŗ©ņĀĢņ¦Ćņ¢┤ņä£ļŖö ņĢł ļÉĀ Ļ▓āņ£╝ļĪ£ ņāØĻ░üļÉ£ļŗż. ņĮöĒśĖĒŖĖ 1ņŚÉņä£ ņ¦łļ¼Ė 4ņØś ņĀÉņłśļŖö ņĢłĻĄ¼Ēæ£ļ®┤ņŚ╝ņāēņ¦ĆņłśņÖĆ ļłłļ¼╝ ļé┤ MMP-9ņØś ļ░£Ēśä ļō▒ĻĖēĻ│╝ļÅä ņ£ĀņØśĒĢ£ ņ¢æņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņŚ¼ ņĢłĻĄ¼ Ēæ£ļ®┤ņØś ņŚ╝ņ”Ø ņłśņżĆĻ│╝ ņŚ░Ļ┤Ćņä▒ņØ┤ ņ׳ņØīņØä ļéśĒāĆļāłļŗż. ņ¦łļ¼Ė 2ņÖĆ ņ¦łļ¼Ė 4 ļ¬©ļæÉ ņĀÉņĢł ļČłĒÄĖĻ░ÉņŚÉ ļīĆĒĢ£ ņŗ¼ļ”¼ ļ░śņØæņØä ĒÅēĻ░ĆĒĢśļŖö ņ£Āņé¼ ĒĢŁļ¬®ņ×äņŚÉļÅä ļČłĻĄ¼ĒĢśĻ│Ā ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņØĖ ļ│ĆņłśļōżņØĆ ņä£ļĪ£ ļŗ¼ļ×ÉļŖöļŹ░, ņØ┤ļŖö ņ¦łļ¼Ė 2Ļ░Ć ņĀÉņĢł ņ¦üņĀäņŚÉ ĒÖśņ×ÉĻ░Ć ļŖÉļü╝ļŖö ļČłĒÄĖĻ░ÉņØä ĒÅēĻ░ĆĒĢśņŚ¼ ĻĘĖ ļ¬®ņĀüņØ┤ ņĀÉņĢł ļŗ╣ņŗ£ņØś ņĀÉņĢłĻ░É ĒÅēĻ░ĆņŚÉ ņŻ╝ņĢłņĀÉņØä ļæÉņŚłĻ│Ā, ņ¦łļ¼Ė 4ļŖö ņĀÉņĢł ļČłĒÄĖĻ░ÉņŚÉ ļīĆĒĢ£ ĒÅēņāüņŗ£ņØś ņŗ¼ļ”¼ņĀü ņŖżĒŖĖļĀłņŖż ņłśņżĆņØä ĒÅēĻ░ĆĒĢśĻĖ░ ļĢīļ¼ĖņŚÉ Ļ▒┤ņä▒ņĢłņØś ņżæņ”ØļÅäņÖĆ ļåÆņØĆ ņāüĻ┤Ćņä▒ņØä ļ│┤ņśĆļŗżĻ│Ā ņāØĻ░üļÉ£ļŗż.
ņ¦łļ¼Ė 5) ņ¦Ćļé£ 4ņŻ╝Ļ░ä ļéśļ”ä ņל ņĀÉņĢłĒĢśņśĆļŗżĻ│Ā ņāØĻ░üĒĢśļŖöņ¦ĆņØś ņĀÉņłśļŖö ļłłļ¼╝ ļé┤ MMP-9ņØś ļ░£Ēśä ļō▒ĻĖēĻ│╝ ņØīņØś ņāüĻ┤ĆĻ┤ĆĻ│äļź╝ ļ│┤ņŚ¼ ņĢłĻĄ¼ Ēæ£ļ®┤ņØś ņŚ╝ņ”ØņØ┤ ļåÆņØäņłśļĪØ ņĢĮņĀ£ ņł£ņØæļÅäĻ░Ć ļé«ņØīņØä ļéśĒāĆļé┤ņŚłļŗż. Ļ▓░ļ¦ēņØś ļ»Ėļ×ĆņØĆ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÖśņ×ÉņŚÉņä£ ņØĖĒä░ļŻ©Ēé©-6, ņØĖĒä░ļŻ©Ēé©-17, ņØĖĒä░ĒÄśļĪĀ Ļ░Éļ¦ł, MMP-9 ļ░£ĒśäņØä ĒżĒĢ©ĒĢ£ ņĢłĻĄ¼ Ēæ£ļ®┤ņØś ņŚ╝ņ”ØĻ│╝ ļÜ£ļĀĘĒĢśĻ▓ī ņŚ░Ļ┤ĆļÉ£ļŗżĻ│Ā ņĢīļĀżņĪīĻ│Ā[32], ņĄ£ĻĘ╝ 2017ļģä TFOS DEWS II ļ”¼ĒżĒŖĖņŚÉ ņØśĒĢśļ®┤ ņĢłĻĄ¼ Ēæ£ļ®┤ņØś ņŚ╝ņ”ØĻ│╝ Ļ▒┤ņĪ░ļŖö ļŗżĒśĢņŗØ ĒåĄĻ░üņłśņÜ®ĻĖ░(polymodal nociceptor)ļź╝ ĒåĄĒĢ£ ļæöĒĢ£ ĒåĄņ”ØĻ│╝ ĻĖ░Ļ│ä ĒåĄĻ░üņłśņÜ®ĻĖ░(mechano-nociceptor)ļź╝ ĒåĄĒĢ£ ļéĀņ╣┤ļĪ£ņÜ┤ ĒåĄņ”ØņØä ņ£Āļ░£ĒĢ£ļŗżĻ│Ā ņĢīļĀżņ¦ä ņé¼ņŗżņØä ĻĘ╝Ļ▒░ļĪ£[33], ņĢłĻĄ¼ Ēæ£ļ®┤ņØś ļ│┤ļŗż ņŗ¼ĒĢ£ ņŚ╝ņ”ØņØ┤ ņĀÉņĢł ņŗ£ ĒåĄĻ░üņłśņÜ®ĻĖ░ ņ×ÉĻĘ╣ņØä ĒåĄĒĢ£ ļČłĒÄĖĻ░ÉĻ│╝ ĻĘĖļĪ£ ņØĖĒĢ£ ļé«ņØĆ ņł£ņØæļÅäļź╝ ņ£Āļ░£Ē¢łļŗżĻ│Ā ņāØĻ░üļÉ£ļŗż.
ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņĢłņĢĮņĀ£ņØś ņĀÉņĢł ņŗ£ ņÖ£ ņ×ÉĻĘ╣Ļ░ÉņØ┤ ļ░£ņāØĒĢśļŖöņ¦ĆņŚÉ ļīĆĒĢ£ ņäĖļČĆ ĻĖ░ņĀäņØĆ ņĢäņ¦üĻ╣īņ¦Ć ņל ņĢīļĀżņ¦ä ļ░ö ņŚåļŗż. ļŗżļ¦ī, ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ņØä ņŚÉļ®ĆņĀ╝ Ēś╝ĒĢ®ņĀ£ ĒśĢĒā£ļĪ£ ļ¦īļōżĻĖ░ ņ£äĒĢ┤ 0.05% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņØīņØ┤ņś© ņŚÉļ®ĆņĀ╝ ņĀÉņĢłņĀ£(Restasis┬«, Allergan)ļŖö ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ņØä Ēö╝ļ¦łņ×Éņ£ĀņŚÉ ļģ╣ņØĖ Ēøä polysorbate 80ņØä Ļ│äļ®┤ĒÖ£ņä▒ņĀ£ļĪ£ ņé¼ņÜ®ĒĢśĻ│Ā, 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖņŚÉļ®ĆņĀ╝ ņĀÉņĢłņĀ£(Ikervis┬«, Santen SAS)ļŖö ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ņØä ņżæņćäĒŖĖļ”¼ĻĖĆļ”¼ņäĖļ”¼ļō£ņŚÉ ļģ╣ņØĖ Ēøä TyloxapolĻ│╝ Poloxamer 188ņØä Ļ│äļ®┤ĒÖ£ņä▒ņĀ£ļĪ£ ņé¼ņÜ®ĒĢśļŖöļŹ░[12], ņØ┤ Ļ│äļ®┤ĒÖ£ņä▒ņĀ£Ļ░Ć ņØ╝ļ¬ģ ņ║Īņé¼ņØ┤ņŗĀ ņłśņÜ®ņ▓┤ļØ╝Ļ│Ā ņĢīļĀżņ¦ä transient receptor potential vanilloid type 1 (TRPV1) ĒåĄĻ░üņłśņÜ®ĻĖ░ļź╝ ņ×ÉĻĘ╣ĒĢśņŚ¼ ņĢłĻĄ¼ Ēæ£ļ®┤ņØś ņ×ÉĻĘ╣Ļ░ÉņØä ņ£Āļ░£ĒĢĀ ņłś ņ׳ļŗż[34]. Ļ│╝Ļ▒░ ĒĢ£ ņŚ░ĻĄ¼ņŚÉņä£ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ļ×½ļō£ ļ¬©ļŹĖņŚÉņä£ TRPV1Ļ│╝ transient receptor potential melastatin 8 ņ▒äļäÉ ņżæ ĒŖ╣Ē׳ TRPV1 ņłśņÜ®ĻĖ░Ļ░Ć ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”ØņŚÉņä£ ĒŖ╣Ē׳ ļŹö Ļ│╝ņןļÉ£ ĒåĄņ”ØņØä ņ£Āļ░£ĒĢ£ļŗżĻ│Ā ņĢīļĀżņ¦ä ņé¼ņŗżņØĆ ņØ┤ļź╝ ļÆĘļ░øņ╣©ĒĢśļ®░[35], ņĢłĻĄ¼ Ēæ£ļ®┤ņØś ļ¦īņä▒ ņŚ╝ņ”ØņØĆ Ļ░üļ¦ēņØś ļ¦Éņ┤ł ņ¦ĆĻ░üņŗĀĻ▓ĮņØä Ļ░Éņ×æņŗ£ĒéżĻ│Ā TRPV1ņØä ļ│ĆĒśĢņŗ£ņ╝£ ņēĮĻ▓ī, ņĀĆņĀłļĪ£ Ēś╣ņØĆ Ļ│╝ņןļÉśĻ▓ī ņ×ÉĻĘ╣ļÉśļŖö ņŗĀĻ▓ĮĻ░Ćņåīņä▒(neuroplastic) ļ│ĆĒÖöļź╝ Ļ░ĆņĀĖņś©ļŗżļŖö ņĀÉ ņŚŁņŗ£ ņāüļīĆņĀüņ£╝ļĪ£ ņŗ¼ĒĢ£ ņŚ╝ņ”ØņØä ļÅÖļ░śĒĢ£ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÖśņ×ÉņŚÉņä£ ņ¦łļ¼Ė 4ņØś ņĀÉņłśĻ░Ć ļŹö ļåÆĻ▓ī ļéśņś© Ļ▓░Ļ│╝ļź╝ ņäżļ¬ģĒĢĀ ņłś ņ׳ļŗż[36].
ņĮöĒśĖĒŖĖ 2ņŚÉņä£ 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ ņĀÉņĢłņĀ£ļź╝ ņĀÉņĢłĒĢśĻ│Ā ņĄ£ņ┤ł ņĢĮ 2.65Ļ░£ņøö ņØ┤Ēøä ņ¦łļ¼Ė 1ņØś ņĀÉņĢł ņ¦üĒøä ļČłĒÄĖĻ░É ņĀÉņłśļŖö ņ£ĀņØśĒĢśĻ▓ī Ļ░ÉņåīĒĢśņśĆĻ│Ā, ņØ┤Ēøä ņČöĻ░ĆņĀüņØĖ 2.61Ļ░£ņøöņØś ņĀÉņĢł ĒøäņŚÉļÅä Ļ░ÉņåīĒĢ£ ļČłĒÄĖĻ░É ņĀÉņłśļŖö ņ£Āņ¦ĆļÉśņŚłļŗż. ļ│Ė ņŚ░ĻĄ¼Ļ░Ć ņäżļ¼Ė ņŗ£Ē¢ēļ¦łļŗż ļ¬©ļōĀ ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÅēĻ░Ć ĒĢŁļ¬®ņØä ļÅÖļ░ś ĒÅēĻ░ĆĒĢśļŖö ņĀäĒ¢źņĀü ņŚ░ĻĄ¼Ļ░Ć ņĢäļŗłĻĖ░ ļĢīļ¼ĖņŚÉ, ņ¦łļ¼Ė ņĀÉņłśĻ░Ć ĒśĖņĀäļÉ£ ņŗ£ņĀÉņŚÉņä£ ļłłļ¼╝ ļé┤ MMP-9 ļ░£Ēśä ļ░Å SICCA ņĢłĻĄ¼Ēæ£ļ®┤ ņŚ╝ņāēņ¦ĆņłśņØś ļ│ĆĒÖöĻ░Ć ļÅÖļ░śļÉśņŚłļŖöņ¦ĆļŖö ĒÖĢņØĖĒĢĀ ņłś ņŚåņŚłļŗż. ļö░ļØ╝ņä£, ņĀÉņĢł ņŗ£ ļČłĒÄĖĻ░ÉņØ┤ ņÖäĒÖöļÉ£ ņØ┤ņ£ĀĻ░Ć ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ņŚ╝ņ”ØņØ┤ ĒśĖņĀäļÉśņ¢┤ņä£ņØĖņ¦Ć ņĢäļŗłļ®┤ ņןĻĖ░Ļ░ä ņĀÉņĢłĒĢśļ®┤ņä£ ĒÖśņ×ÉĻ░Ć ņĢĮņĀ£ ņĀÉņĢł ņŗ£ņØś ļČłĒÄĖĻ░ÉņŚÉ ĒĢÖņŖĄļÉśĻ│Ā ņĀüņØæņØ┤ ļÉ£ Ļ▓āņØĖņ¦Ćļź╝ ļ│Ė ņŚ░ĻĄ¼ņŚÉņä£ļŖö ĻĄ¼ļČäĒĢĀ ņłś ņŚåļŗż. Ē¢źĒøä ļ│┤ļŗż ļ¦ÄņØĆ ņłśņØś ĒÖśņ×É ņłśļź╝ ļīĆņāüņ£╝ļĪ£ ņØ╝ņĀĢĒĢ£ Ļ░äĻ▓®ņ£╝ļĪ£ ņäżļ¼ĖņØä ņŗ£Ē¢ēĒĢśĻ│Ā ņĢłĻ│╝ņĀü Ļ▓Ćņé¼ļź╝ ļÅÖļ░ś ņŗ£Ē¢ēĒĢśļÅäļĪØ ņäżĻ│äļÉ£ ņĀäĒ¢źņĀüņØĖ ņŚ░ĻĄ¼ļź╝ ņŗ£Ē¢ēĒĢ£ļŗżļ®┤ ņČöĻ░ĆņĀüņØ┤Ļ│Ā ļ│┤ļŗż ļ¬ģĒÖĢĒĢ£ Ļ▓░Ļ│╝ļź╝ ļ¬©ņāēĒĢ┤ļ│╝ ņłś ņ׳Ļ▓Āļŗż.
ļ│Ė ņŚ░ĻĄ¼ļŖö EDSQļØ╝ļŖö ņל ņĢīļĀżņĀĖ ņ׳ņ¦Ć ņĢŖņØĆ ņäżļ¼Ėņ¦Ć ņżæ ņØ╝ļČĆļź╝ ņ░©ņÜ®ĒĢśņśĆĻ│Ā ņäżļ¼Ėņ¦Ć ļé┤ Ļ░ü ĒĢŁļ¬®ļōżņØ┤ ņĢłņĢĮ ņĀÉņĢł ņŗ£ ļČłĒÄĖĻ░ÉņØś ņóģļźśļź╝ ĻĄ¼ļČäĒĢ┤ņä£ ļ│┤ņŚ¼ņŻ╝Ļ▒░ļéś ņł£ņØæļÅäņÖĆ ļ╣äņł£ņØæļÅäļź╝ ņäĖļČĆņĀüņ£╝ļĪ£ ļéśĒāĆļé┤ņ¦Ć ļ¬╗ĒĢśļŖö ĒĢ£Ļ│äņĀÉņØ┤ ņ׳ļŗż. ĒĢśņ¦Ćļ¦ī, ņĢłĻĄ¼Ļ▒┤ņĪ░ņ”Ø ĒÅēĻ░Ć ņäżļ¼Ėņ¦ĆņÖĆļŖö ļŗżļź┤Ļ▓ī ņĢłņĢĮ ņĀÉņĢłĻ░ÉņØä ĒÅēĻ░ĆĒĢĀ ņłś ņ׳ļŖö ņäżļ¼Ėņ¦ĆļŖö ļŗżņ¢æĒĢśņ¦Ć ņĢŖļŗż. ļśÉĒĢ£, ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņĢłņĢĮņØś ņĀÉņĢłĻ░É ļ░Å ņł£ņØæļÅäņŚÉ ļ»Ėņ╣śļŖö ņśüĒ¢źļĀź ņØĖņ×ÉņŚÉ ļīĆĒĢ┤ ņØ┤ņĀ£Ļ╗Å ĻĄŁļé┤ņÖĖ ņŚ░ĻĄ¼ļÉ£ ļ░öļŖö ņŚåņŚłļŗż. ņØ┤ņŚÉ ĻĄŁļé┤ņŚÉņä£ 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ ņĀÉņĢłņĀ£ņØś ņĀÉņĢł ņ¦üĒøä ļČłĒÄĖĻ░ÉĻ│╝ ņØ┤ļĪ£ ņØĖĒĢ£ ļ╣äņł£ņØæņØ┤ ņŗżņĀ£ ņ×äņāü ĒÖśĻ▓ĮņŚÉņä£ ņżæņÜöĒĢ£ ņØ┤ņŖłĻ░Ć ļÉśĻ│Ā ņ׳ļŖö ņ¦ĆĻĖł, ļ│Ė ņŚ░ĻĄ¼ņØś Ļ▓░Ļ│╝ļź╝ ņĀüņĀłĒĢśĻ▓ī ĒÖ£ņÜ®ĒĢ£ļŗżļ®┤ 0.1% ņé¼ņØ┤Ēü┤ļĪ£ņŖżĒżļ”░ ņ¢æņØ┤ņś© ļéśļģĖ ņŚÉļ®ĆņĀ╝ ņĀÉņĢłņĀ£ņØś ņĀÉņĢłĻ░ÉņŚÉ ļīĆĒĢ£ ĒÖśņ×Éļ│ä ļ¦×ņČż ņäżļ¬ģĻ│╝ ņł£ņØæļÅä Ē¢źņāüņŚÉ ļÅäņøĆņØä ņżä Ļ▓āņ£╝ļĪ£ ĻĖ░ļīĆļÉ£ļŗż.
Figure┬Ā1.
Questionnaire to evaluate the ocular discomfort and compliance of eye drops in this study. The questions of this questionnaire are reproduced in Korean and are adapted from the originally pre-established Eye-drop satisfaction questionnaire (EDSQ).

Figure┬Ā2.
Analysis of the serial change of the scores of questions 1 to 7 with the use of 0.1% cyclosporine A cationic nanoemulsion instillation in cohort 2. (A-D) Serial scores of questions 1 to 4 over the three times of periodic questionnaire in the dimension ŌĆśconcerns about treatmentŌĆÖ to verify the ocular discomfort on instillation. (E-G) Serial scores of questions 5 to 7 over the three times of periodic questionnaire in the dimension ŌĆśself-declared complianceŌĆÖ to verify the drug compliance. Q = question. *p-values were calculated using Wilcoxon signed rank test, p < 0.05.
Table┬Ā1.
Demographics and parameters for dry eye disease in cohort 1
Table┬Ā2.
Demographics in cohort 2
| Variable | Value (Cohort 2) |
|---|---|
| Total number of patients/eyes | 34 |
| Age (years, mean ┬▒ standard deviation) | 54.5 ┬▒ 15.7 |
| Male/female (female, %) | 11/23 (68) |
Table┬Ā3.
Correlations of demographics and parameters for dry eye disease with scores of questions in ŌĆśconcerns about treatmentŌĆÖ dimension of the questionnaire in cohort 1
| Variable* |
Dimension: concerns about treatment |
|||||
|---|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | |||
| Score | 2.59 ┬▒ 1.85 | 2.05 ┬▒ 2.01 | 2.18 ┬▒ 2.02 | 1.69 ┬▒ 1.91 | ||
| vs. Age (years) | rs coefficient | -0.143 | -0.101 | -0.138 | -0.247 | |
| p-value | 0.261 | 0.430 | 0.277 | 0.049ŌĆĀ | ||
| vs. Duration of eyedrops (months) | rs coefficient | -0.122 | -0.389 | -0.029 | -0.199 | |
| p-value | 0.337 | 0.002ŌĆĪ | 0.821 | 0.115 | ||
| vs. MG expressibility (Gr) | rs coefficient | 0.063 | -0.005 | 0.182 | 0.117 | |
| p-value | 0.613 | 0.970 | 0.140 | 0.345 | ||
| vs. Meibum quality (Gr) | rs coefficient | -0.179 | -0.041 | 0.172 | -0.120 | |
| p-value | 0.148 | 0.740 | 0.163 | 0.334 | ||
| vs. Tear secretion (mm) | rs coefficient | 0.083 | 0.289 | 0.174 | 0.048 | |
| p-value | 0.569 | 0.042ŌĆĀ | 0.234 | 0.743 | ||
| vs. Corneal sensitivity (mm) | rs coefficient | -0.127 | 0.122 | -0.046 | -0.005 | |
| p-value | 0.423 | 0.441 | 0.771 | 0.977 | ||
| vs. Corneal erosion score | rs coefficient | 0.027 | -0.025 | 0.297 | 0.204 | |
| p-value | 0.826 | 0.843 | 0.015ŌĆĀ | 0.098 | ||
| vs. OSS | rs coefficient | 0.265 | 0.107 | 0.009 | 0.298 | |
| p-value | 0.079 | 0.485 | 0.952 | 0.047ŌĆĀ | ||
| vs. Tear MMP-9 (Gr) | rs coefficient | -0.099 | 0.140 | -0.106 | 0.287 | |
| p-value | 0.467 | 0.303 | 0.436 | 0.032ŌĆĀ | ||
| vs. OSDI score | rs coefficient | 0.246 | 0.154 | 0.228 | 0.210 | |
| p-value | 0.175 | 0.401 | 0.209 | 0.249 | ||
|
|
||||||
| Variable┬¦ | Q1 | Q2 | Q3 | Q4 | ||
|
|
||||||
| Sex | Male | Score | 1.38 ┬▒ 2.00 | 2.13 ┬▒ 2.17 | 1.38 ┬▒ 1.77 | 1.13 ┬▒ 1.89 |
| Female | 2.90 ┬▒ 1.70 | 2.03 ┬▒ 2.01 | 2.39 ┬▒ 2.06 | 1.84 ┬▒ 1.92 | ||
| p-value | 0.048ŌĆĀ | 0.827 | 0.269 | 0.259 | ||
Table┬Ā4.
Correlations of demographics and parameters for dry eye disease with scores of questions in ŌĆśself-declared complianceŌĆÖ dimension of the questionnaire in cohort 1
| Variable* |
Dimension: self-declared compliance |
||||
|---|---|---|---|---|---|
| Q5 | Q6 | Q7 | |||
| Score | 3.77 ┬▒ 1.60 | 1.15 ┬▒ 1.50 | 1.41 ┬▒ 1.92 | ||
| vs. Age (years) | rs coefficient | 0.300 | -0.103 | -0.050 | |
| p-value | 0.064 | 0.533 | 0.764 | ||
| vs. Duration of eyedrops (months) | rs coefficient | 0.166 | -0.142 | 0.028 | |
| p-value | 0.313 | 0.389 | 0.867 | ||
| vs. MG expressibility (Gr) | rs coefficient | -0.094 | -0.107 | -0.050 | |
| p-value | 0.450 | 0.391 | 0.690 | ||
| vs. Meibum quality (Gr) | rs coefficient | <-0.001 | -0.065 | -0.181 | |
| p-value | 0.998 | 0.599 | 0.142 | ||
| vs. Tear secretion (mm) | rs coefficient | 0.053 | -0.189 | 0.018 | |
| p-value | 0.714 | 0.190 | 0.900 | ||
| vs. Corneal sensitivity (mm) | rs coefficient | -0.251 | 0.231 | 0.194 | |
| p-value | 0.109 | 0.141 | 0.218 | ||
| vs. Corneal erosion score | rs coefficient | 0.050 | 0.110 | -0.087 | |
| p-value | 0.690 | 0.375 | 0.483 | ||
| vs. OSS | rs coefficient | -0.203 | -0.038 | -0.096 | |
| p-value | 0.181 | 0.803 | 0.532 | ||
| vs. Tear MMP-9 (Gr) | rs coefficient | -0.320 | 0.178 | 0.149 | |
| p-value | 0.016ŌĆĀ | 0.190 | 0.273 | ||
| vs. OSDI score | rs coefficient | -0.001 | -0.052 | 0.007 | |
| p-value | 0.994 | 0.776 | 0.968 | ||
|
|
|||||
| Variable┬¦ | Q5 | Q6 | Q7 | ||
|
|
|||||
| Sex | Male | Score | 3.50 ┬▒ 2.20 | 0.38 ┬▒ 0.74 | 0.75 ┬▒ 1.75 |
| Female | 3.84 ┬▒ 1.44 | 1.36 ┬▒ 1.58 | 1.58 ┬▒ 1.95 | ||
| p-value | 0.955 | 0.092 | 0.222 | ||
REFERENCES
1) Craig JP, Nichols KK, Akpek EK, et al. TFOS DEWS II Definition and Classification Report. Ocul Surf 2017;15:276-83.


2) Bartlett JD, Keith MS, Sudharshan L, Snedecor SJ. Associations between signs and symptoms of dry eye disease: a systematic review. Clin Ophthalmol 2015;9:1719-30.



3) McDonald M, Patel DA, Keith MS, Snedecor SJ. Economic and humanistic burden of dry eye disease in Europe, North America, and Asia: a systematic literature review. Ocul Surf 2016;14:144-67.


6) Strong B, Farley W, Stern ME, Pflugfelder SC. Topical cyclosporine inhibits conjunctival epithelial apoptosis in experimental murine keratoconjunctivitis sicca. Cornea 2005;24:80-5.


7) Baudouin C, de la Maza MS, Amrane M, et al. One-year efficacy and safety of 0.1% cyclosporine a cationic emulsion in the treatment of severe dry eye disease. Eur J Ophthalmol 2017;27:678-85.



8) Baudouin C, Figueiredo FC, Messmer EM, et al. A randomized study of the efficacy and safety of 0.1% cyclosporine A cationic emulsion in treatment of moderate to severe dry eye. Eur J Ophthalmol 2017;27:520-30.



9) Leonardi A, Van Setten G, Amrane M, et al. Efficacy and safety of 0.1% cyclosporine A cationic emulsion in the treatment of severe dry eye disease: a multicenter randomized trial. Eur J Ophthalmol 2016;26:287-96.


10) Byun YS, Rho CR, Cho K, et al. Cyclosporine 0.05% ophthalmic emulsion for dry eye in Korea: a prospective, multicenter, open-label, surveillance study. Korean J Ophthalmol 2011;25:369-74.



11) Daull P, Lallemand F, Garrigue JS. Benefits of cetalkonium chloride cationic oil-in-water nanoemulsions for topical ophthalmic drug delivery. J Pharm Pharmacol 2014;66:531-41.


12) Lallemand F, Schmitt M, Bourges JL, et al. Cyclosporine A delivery to the eye: a comprehensive review of academic and industrial efforts. Eur J Pharm Biopharm 2017;117:14-28.


13) Lallemand F, Daull P, Benita S, et al. Successfully improving ocular drug delivery using the cationic nanoemulsion, novasorb. J Drug Deliv 2012;2012:604204.



14) Daull P, Lallemand F, Philips B, et al. Distribution of cyclosporine A in ocular tissues after topical administration of cyclosporine A cationic emulsions to pigmented rabbits. Cornea 2013;32:345-54.


15) Lallemand F, Felt-Baeyens O, Besseghir K, et al. Cyclosporine A delivery to the eye: a pharmaceutical challenge. Eur J Pharm Biopharm 2003;56:307-18.


16) de Oliveira RC, Wilson SE. Practical guidance for the use of cyclosporine ophthalmic solutions in the management of dry eye disease. Clin Ophthalmol 2019;13:1115-22.


17) Labetoulle M, Leonardi A, Amrane M, et al. Persistence of efficacy of 0.1% cyclosporin a cationic emulsion in subjects with severe keratitis due to dry eye disease: a nonrandomized, open-label extension of the SANSIKA study. Clin Ther 2018;40:1894-906.


19) Regnault A, Viala-Danten M, Gilet H, Berdeaux G. Scoring and psychometric properties of the Eye-Drop Satisfaction Questionnaire (EDSQ), an instrument to assess satisfaction and compliance with glaucoma treatment. BMC Ophthalmol 2010;10:1.



20) Nordmann JP, Denis P, Vigneux M, et al. Development of the conceptual framework for the Eye-Drop Satisfaction Questionnaire (EDSQ) in glaucoma using a qualitative study. BMC Health Serv Res 2007;7:124.



21) Pflugfelder SC, Tseng SC, Sanabria O, et al. Evaluation of subjective assessments and objective diagnostic tests for diagnosing tear-film disorders known to cause ocular irritation. Cornea 1998;17:38-56.


22) Tomlinson A, Bron AJ, Korb DR, et al. The international workshop on meibomian gland dysfunction: report of the diagnosis subcommittee. Invest Ophthalmol Vis Sci 2011;52:2006-49.



23) Park JY, Kim BG, Kim JS, Hwang JH. Matrix metalloproteinase 9 point-of-care immunoassay result predicts response to topical cyclosporine treatment in dry eye disease. Transl Vis Sci Technol 2018;7:31.

24) Lemp MA. Report of the national eye institute/industry workshop on clinical trials in dry eyes. CLAO J 1995;21:221-32.

25) Whitcher JP, Shiboski CH, Shiboski SC, et al. A simplified quantitative method for assessing keratoconjunctivitis sicca from the Sj├Čgren's Syndrome International Registry. Am J Ophthalmol 2010;149:405-15.


26) Epitropoulos AT, Therattil A, Periman LM, Rosenberg ED. Improving tolerance and compliance with topical immunomodulators using micro-emulsion lipid layer artificial tears. Clin Ophthalmol 2020;14:1921-9.


27) Perry HD, Solomon R, Donnenfeld ED, et al. Evaluation of topical cyclosporine for the treatment of dry eye disease. Arch Ophthalmol 2008;126:1046-50.


28) Sall K, Stevenson OD, Mundorf TK, Reis BL. Two multicenter, randomized studies of the efficacy and safety of cyclosporine ophthalmic emulsion in moderate to severe dry eye disease. CsA Phase 3 Study Group. Ophthalmology 2000;107:631-9.


29) Li W, Graham AD, Lin MC. Understanding ocular discomfort and dryness using the pain sensitivity questionnaire. PLoS One 2016;11:e0154753.



30) Stern ME, Gao J, Siemasko KF, et al. The role of the lacrimal functional unit in the pathophysiology of dry eye. Exp Eye Res 2004;78:409-16.


31) Roszkowska AM, Colosi P, Ferreri FM, Galasso S. Age-related modifications of corneal sensitivity. Ophthalmologica 2004;218:350-5.


32) Yang S, Lee HJ, Kim DY, et al. The use of conjunctival staining to measure ocular surface inflammation in patients with dry eye. Cornea 2019;38:698-705.


33) Belmonte C, Nichols JJ, Cox SM, et al. TFOS DEWS II pain and sensation report. Ocul Surf 2017;15:404-37.



34) Lilja J, Lindegren H, Forsby A. Surfactant-induced TRPV1 activity--a novel mechanism for eye irritation? Toxicol Sci 2007;99:174-80.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print